Chapter 22
The application of basic concepts of Zeta Potential to Cardiovascular Disease.
from
Control of Colloid Stability through Zeta Potential
With this closing chapter on its relationship
to cardiovascular disease
Vol. I
by
Thomas M. Riddick
Consulting Engineer and Chemist, Thomas M. Riddick and Associates,
New York, New York — and Technical Director
Copyright © 1968 by Thomas M. Riddick
|
Zeta Potential represents a basic law of Nature, and it plays a vital role in all forms of plant and animal life.
It is the force that
maintains the discreteness of the billions of circulating cells, which nourish the organism.
The stability of simple inorganic man–made systems is governed by these same laws.
The relevance and application of these principles is the subject of this book.
|
INTRODUCTION
In the past it may have been considered questionable taste to include personal experiences in a technical book of this sort. If for some valid reason this must be done, one technique has been to retreat to the anonymity of the third person. The writer is purposely departing from this precedent, and presents this final chapter concerning heart disease in the first person. This is because my interest in this subject is more than academic. It is first hand, personal and intimate — and stems from the fact that during the past twelve years it has become increasingly evident that I have a cardiovascular condition. I do not mean to be dramatic, but the simple fact is, I am not ready or willing to die at the age of sixty. And I deeply resent the attitude of complete indifference on the part of Federal authorities toward correcting the conditions that I have reasonable proof are correctable causative factors in heart disease.
EARLY EXPERIENCES WITH ANGINA, PAROXYSMAL TACHYCARDIA,
AND PREMATURE VENTRICULAR CONTRACTIONS
I will relate my personal experience with cardiovascular disease only in sufficient detail to justify, or give reasons for, the action I took.
Unless one experiences a heart attack of some severity, involving hospitalization, it is difficult to pin–point just when the condition started. Mine began with anginal pains, and the date appearing on my nitroglycerine prescription is March 7, 1955, or 12 years ago! The episodes of acute pain were not frequent or severe — just a few each year. I recall making several trips to the local hospital after midnight.
Paroxysmal tachycardia, another manifestation of cardiovascular disease, soon appeared. It was reasonably advanced by August 1957, because I can remember a severe attack at that time and the circumstances attending it.
Two years ago, my comprehensive medical examination read in part: "Ventricular premature contractions in all leads — Low voltage QRS in the limb leads." The interpretation stated: "The changes noted in your electrocardiogram are not important. We consider them to be within the normal limits."
I verified the premature ventricular contractions* (PVC's) over a period of several weeks by placing my thumb and forefinger on my carotid arteries.
[ * Many types of cardiovascular disease are manifest in the early stages as "moderate to significant" intravascular coagulation, and in advanced stages as "heavy to very heavy" coagulation. In this latter stage, PVC's or malfunctional heartbeats, which throw virtually no blood, may range from 1 to as high as 50% of total heartbeats. The medical profession recognizes many different types of heart disease. These were reviewed briefly for us by a physician and are included as Appendix II. ]
My pulse "skipped a beat" about once every four to six beats. I did not then, nor do I now, consider such malfunction is "not important"; or "within normal limits." A PVC is very important to me. Any atypical beat is important to me, and any malfunction* is distressing to me.
[ * I have discussed the importance of malfunctional heartbeats, which throw no blood with a number of competent cardiologists and general practitioners. The ones I consulted consider the PVC to be extremely important. However, I understand that some medical men consider it unimportant. In my own field, we would not consider a malfunctioning valve in a chemical feed pump as normal, nor would we view the periodic failure of an electrical control device as unimportant. ]
Let me emphasize that I do not blame my physician for this state. While I had long been under his general care and was considered "reasonably healthy," there was nothing he could have done to forestall this development, or even foresee it. After all, every day, often without warning cardiologists and general practitioners die of heart disease in their 40's, 50's and 60's.*
[ * It seems significant that Representative John E. Fogarty of Rhode Island, who had suffered a heart attack in 1953, literally devoted his life to raising funds for expanding the cardiovascular research of the NIH. In 1966, in recognition of the extent of his recovery he was decorated by President Johnson as the "Heart of the Year." He died January 13, 1967, at the age of 53 of a heart attack. In 1965, the head cardiologist of a leading New York hospital died at the age of 43; and the pathologist of the hospital that had long furnished the writer with blood for research, died at the age of 45 — both from heart attacks. These and many other such cases point to the actual paucity of definitive knowledge of cardiovascular disease. ]
From a scientific point of view, it is difficult to reconcile this with the tremendous amount of research money and effort being expended without seriously questioning the validity and efficacy of the present approach.
Strictly from a physical chemist's point of view, one may seriously question the validity of the chemistry and the rationale presently being applied it was intimately associated with morbidity and to the basic problems of cardiovascular disease.
Returning to my personal case, my physician apparently could not correct this malfunction or prevent this tachycardia. And to state that it was "normal," from his standpoint was true. Unfortunately, it is presently normal in the United States for 55 out of 100 persons to die of some form of cardiovascular disease. It would seem that when a person is experiencing one malfunctional heartbeat in five, with unpredictable paroxysmal tachycardia, it is now accepted practice to simply await a more severe "heart attack" and then hospitalize the patient and place him on heparin, dicumaroll, coumadin, or a similar drug.
Heparin is an excellent dispersing agent, but is apparently too large a polymer to diffuse from the stomach into the bloodstream. Therefore, it must be administered by hypodermic injection into the fat of the abdomen, twice daily, for "life." But with dicumaroll and other similar orally administered drugs, results apparently have not been so successful.
DECISION TO EMPLOY ZETA POTENTIAL
— BUT ONLY WITHIN MY OWN FIELD OF PROFESSIONAL COMPETENCE
I began continuous research on Zeta Potential in 1956, and within two years was convinced that proper development of this subject must lead to the elucidation and control of cardiovascular disease. But much valuable time was then lost in trying to convince the research departments of two of our oldest and largest pharmaceutical houses. They thought the odds were too great. As one put it: "How can we successfully compete with $400,000,000 annual federally subsidized research?" My only alternative was to continue my work alone, and unaided. But the more I worked with systems (from industrial concerns) and blood (from a local hospital), the more convinced I became that blood stability followed all the rules of colloid stability. And too, I became more inclined to believe that the majority of serious cardiovascular disease
[ except hypertension ]
must stem primarily from intravascular coagulation.
I was not then aware that Melvin Knisely and Edward Bloch (and associates), more than ten years earlier, had explored intravascular coagulation and "blood sludge" extensively.
[ See Refs. 11-16 and 11-47. ]
They had shown it was widespread in human beings and could be induced in test animals. They also revealed that, in extreme, it was intimately associated with morbidity and death. After studying Knisely's reprints, I realized that while I did not actually know these facts, I had long felt that the conditions he described must exist.
When I realized that my heart was seriously malfunctioning, and that my own physician was apparently unable to prevent or control the situation, I decided to put into practice immediately my basic findings in Physical Chemistry and Zeta Potential. I did not wish to await that "hospitalizing" heart attack — I wanted to prevent it. And the prospect of two heparin injections a day for the rest of my life did not appeal to me. Moreover, I wanted not only to eliminate my malfunctional heartbeats, but to learn the nature of and prevent the paroxysmal tachycardia that drove my heart from 80 to 180 beats per minute.
I also resolved that I would attempt to solve this problem strictly within the confines of my own field of professional competence, where I am duly qualified and professionally licensed. For thirty years, I have headed my own firm, as an Engineer and Chemist, engaged in the design, operation and supervision of municipal and industrial Waterworks and Waste Treatment Plants. Moreover, I have continuously operated a laboratory devoted to research, as well as routine examinations of water.
With regard to possible methods of applying Zeta Potential to the problem of intravascular coagulation, it seemed to us that dispersion of the blood system and prevention of agglomerates was the crux of the matter. Therefore, one logical approach would be to drink only distilled (rather than tap) water, and to add to this suitable electrolytes of a dispersing nature. The problem at the outset seemed to be one of Physical Chemistry — not Physiology. We believed that the use of drugs, per se, would be the wrong approach.
To launch this program, we replaced our old oscilloscope with the Tektronix described, and designed and built a "Sclerascope" for horizontally viewing (with the stereoscopic microscope) the blood vessels of the eye. This enabled us to view coagulation. We then developed a system for appraising the extent of intravascular coagulation (IVC). Finally, we gave detailed consideration to which of the safe electrolytes would be the most appropriate, and in what quantities they should be used.
The reader will view my decision to take matters in my own hands in a manner which will, of course, reflect the reader's own background. Some may interpret my action as "a chemist trying to practice medicine." My "medical background" which, by the way, is "considerable," is pertinent at this point.
My father was a country doctor during the horse–and–buggy days at the turn of the century. This meant long hours, and surgery on short notice. In fact, in my native Eastern North Carolina in those days, a door placed on the kitchen table was standard for surgery. My father was the nineteenth physician on my paternal side, which therefore goes back to the pre–Pasteur era when the basic cause of microbial disease was as unknown as the cause of heart disease today. My father died of a kidney infection in 1907, at the age of 53.
From age 12 to 16, because of my early interest in medicine, I served after school as a "Model–T driver and surgical assistant" to Dr. Thomas Carter. He was a country doctor — and gave me every encouragement. While I ultimately chose chemistry and engineering, my early experience with Dr. Carter spared me any sense of awe of medicine that I might later have developed.
Returning to the problem of my malfunctioning heart, I will simply state that by applying certain pertinent basic principles of Zeta Potential and Physical Chemistry to blood stability, I was able to eliminate my malfunctional beats — and also gain insight into some of the vagaries of paroxysmal tachycardia. (It may well be that this information will offer the first logical clue as to its origin.)
A few stories of my father's practice and sense of humor are still alive after sixty years. One of his first cases after medical school was a local woman who had become demented with pain, and addicted to drugs. When he saw her she was considered a "hopeless case" by the four local physicians, who scoffed at my father's diagnosis of "tumor of the womb." And so, he publicly stated that on a certain Wednesday at 3 o'clock he would remove the tumor, and he predicted it would weigh over a pound. In his announcement, he invited the four doctors to be present, and, under the circumstances, they could not refuse. As it developed, the tumor weighed almost two pounds. Over my mother's vigorous protestations, my father had it brought in for the "appraisal and delectation" of his confreres on a large platter intended for serving a roast suckling pig. Ultimately, the woman recovered her senses and no longer required morphine. Three of these physicians became my father's closest friends; one never spoke to him again. Medicine has come a long way since then. But the dedicated physicians of today are just as dedicated as they were then; they are just harder to find. And of course, such professional rivalry no longer exists.
I do not delude myself that I have affected a "complete cure" of my cardiovascular disorder. Without doubt, my heart is (and has long been) infarcted and, from the standpoint of irreversible physicochemical sequences, I do not believe infarction can be eradicated. Within the limits of the present knowledge of chemistry, it would seem that infarction represents a structural defect and permanent damage.
What I have done, in a physicochemical sense, is significantly diminish my former high degree of intravascular coagulation through slight, but appropriate, control of the Zeta Potential of my blood system. I have, as far as is presently possible, only restored it to its normal state; not basically changed it. My red cells (and the other formed elements) are thus freed to a significant extent from the abnormal, encompassing polymers and forces, which bound them into aggregates. Now they are better able to fulfill their required functions of oxygen transport and removal of carbon dioxide and metabolic end products. However, I believe of much greater importance than improved ability of the red cell to transport oxygen, is its increased ability to more freely transmit this oxygen to the countless cells that constitute every organ of the body, including the heart. Therefore, the connotation is much broader than simple anoxemia.
It is a sobering thought that by the simple expedient of ingesting distilled water containing appropriate electrolytes, my malfunctioning heartbeats were corrected and my paroxysmal tachycardia significantly relieved. How much "margin of safety" now exists is unknown. But whatever it is, I believe it would be considerably more if I had known how to correct this condition at its start, ten years ago. It is statistically possible for me to die, even today, from a "heart condition." But surely the probability of premature death is now greatly lessened.
OBSERVATIONS CONCERNING KIDNEY FUNCTIONS,
BLOOD ELECTROLYTES,
AND THE DAILY INTAKE OF WATER AND DISSOLVED MINERAL SALTS
From the point of view of physical chemistry, the writer will now develop the sequence of the cause, prevention and control of cardiovascular disease as it presently appears.
As a country boy, I was impressed by the rapid transfer of liquid from the stomach to the bladder in "watermelon season," and also the quick transfer of asparagus. Later, I was further impressed by the even more rapid transfer of beverage alcohol from the stomach to the blood–stream and brain.
From 1825 to 1835, William Beaumont produced visual evidence that water admitted to the stomach is always absorbed within a few minutes. This is an interesting story and deserves retelling. On June 6, 1822, at Fort Mackinac, Michigan (near the Canadian border), an incident occurred that was to result in some of the most bizarre research ever recorded in medical history. On that date, William Beaumont, a young surgeon with the U.S. Army, treated Alexis St. Martin, an 18–year–old Canadian, for a shotgun–blast at three–foot range. The charge entered just below the left breast, tearing off a large portion of the side . . . . the ribs fractured and openings were made into the cavities of the chest and abdomen (about 2½" diameter) through which protruded portions of the lungs and stomach, much lacerated and burnt." These openings healed peripherally, and were closed internally by a "flap valve" of tissue (actually the inner coats of the stomach), which could be readily pushed aside, enabling ready access and direct view of the stomach.
St. Martin recovered and became the servant of Beaumont, who performed innumerable digestion experiments on him for ten years, beginning May, 1825.*
[ * Experiments and Observations on the Gastric Juice and the Physiology of Digestion — William Beaumont, M.D. — Original Edition 1833 — Dover Publications, 1959. ]
For experiments in vitro, Beaumont would insert a tube through the hole into the stomach several times each week, and draw off about 1½ ounces of "pure gastric juice." We quote Beaumont:
On pressing down the valve when the stomach is full, the contents flow out copiously. When the stomach is nearly empty, and quiescent, the interior of the cavity may be examined to the depth of five or six inches, if kept distended by artificial means; and the food and drinks may be seen entering it, if swallowed at this time, through the ring of the esophagus ... water, ardent spirits, and most other fluids are not affected by the gastric juice, but are passed from the stomach soon after they have been received.
Experiment 68. At 9 o'clock P.M. . . . St. Martin having eaten nothing since 2 o'clock, and feeling quite hungry, I put into the stomach, at the aperture, eight ounces of beef and barley soup, introduced gently through a tube, with a syringe, lukewarm. It caused no unpleasant sensation, but allayed the sense of hunger. It satisfied the appetite; and he said he had no desire to eat. At 10 o'clock, he said he felt a little hungry again, and ate eight ounces more of the same kind of soup, which had a similar effect as the other.
Experiment 71. At 1 o'clock P.M. . . . St. Martin complaining of being quite hungry, I put into the stomach at the aperture, twelve raw oysters, more than middling size. The sensation was allayed, and the appetite satisfied, the same as if swallowed. He was not hungry again till half after 4 o'clock, when he ate a dozen more of the same kind of oysters, with bread. At 10 o'clock P.M., stomach empty and clean. Weather damp and rainy. Wind N.E. and brisk. Temperature of the stomach, 99½°.
Experiment 114. March 27. At 9 o'clock, 15–mins.
A.M., he breakfasted on fresh broiled fish (Flounder), bread and coffee, and kept exercising moderately. 11 o'clock, stomach half empty — pulp of bread only appeared. 11 o'clock, 30 mins., particles of fish and bread still to be seen in the stomach. 1 o'clock P.M., stomach entirely clear of food. Temperature 101°.
Beaumont showed that gastric juice, either in situ or in a warmed test tube will digest foodstuffs. In situ, digestion periods ranged from 1 to 4 hours, depending upon the nature and maceration of the food. Average digestion time was about 3 hours. He also showed that even the bone of a hog's rib would be digested in 21 days.
Beaumont's views on spiritous liquors are also of interest:
Diseased appearances . . . have frequently presented themselves, in the course of my experiments and examinations . . . They have generally, but not always, succeeded to some appreciable cause. Improper indulgence in eating and drinking, has been the most common precursor of these diseased conditions of the coats of the stomach. The free use of ardent spirits, wine, beer, or any intoxicating liquor, when continued for some days, has invariably produced these morbid changes.
St. Martin lived to an old age, and was buried in Canada in a grave eight feet deep. Apparently he didn't want any further experimentation on his stomach.
Now to return to the subject of the rapid absorption of liquids from the stomach. It would seem that this absorption (surely in the case of alcohol) is directly into the blood stream. Thus the blood is very slightly and temporarily diluted (for a period of perhaps 10 to 30 minutes) each time we drink liquids such as water, tea or coffee. These liquids carry a small intake of mineral salts, but it is a very small percentage of our daily mineral intake. Tap water usually has a range of 150 to 1,000 micromhos;*
[ * An eight–oz. glass of water contains 250 ml. At 1000 micromhos, it would be equivalent to 0.12 grams of sodium chloride. Some physiologists hold that liquids such as water are significantly increased in mineral salts before absorption to the blood stream. Beaumont seems to have felt that water absorbed almost instantly, and that gastric juice was never found in the stomach except when food was present. ]
milk (an exception to this rule) 5,000 to 6,000 micromhos; coffee and tea, 1,000 to 1,500 micromhos; beer, 1,700 to 2,500 micromhos. Alcoholic beverages are low to virtually absent in mineral solids content. Foodstuffs have a range that will be considered later in detail, but they represent a mineral intake approximating 18 grams per day.
Goostray and Schwenck**
[ ** A Textbook of Chemistry, Eighth Edition, 1961 — Stella Goostrav and J. Rae Schwenck — The Macmillan Co., New York, N. Y. ]
list the daily intake of sodium in the diet as 2,300 to 6,000 mg. This is equivalent to 5.9 to 12.7, or an average of 9.3 (say 10) grams of NaCl per day. They state that when salt is "not used at the table," the intake is reduced to 1,500 to 2,600 mg of Na, which is equivalent to 3.8–6.1, or an average of 5.0 grams of NaCl. This would imply that of 10 grams daily intake of sodium chloride, one–half represented salt "used at the table." The writer cannot accept this value, and it would seem that two different sets of data might inadvertently have been employed, or by "table use" they included salt employed in cooking.
But the exactness of the figure is not too important here. They also state that low–sodium diets restrict sodium to 585–780 mg/day— equivalent to 1.5–2.0 grams of sodium chloride per day.
|
TABLE NO. 24
|
|---|
|
Range
|
Average
|
|---|
1.) Water Input, Liquid +
Foodstuffs (liters)
|
1.5–3.0
|
(say) 2.0
|
|---|
|
a) Drink, about 50%
|
—
|
1.0
|
|---|
|
b) Food, about 50%
|
—
|
1.0
|
|---|
|
|
|
2.) Water Output, liters
|
|
a) Insensible and vaporization
|
1.0–1.20
|
1.1
|
|---|
|
b) Urine
|
0.80–1.00
|
0.9
|
|---|
|
|
|
Ave. 2.0
|
|---|
|
|
3.) Input and output of
Mineral Salts
|
|
a) Sodium Chloride
|
5–14.5
|
9.7
|
|---|
|
b) Potassium (as chloride)
|
3.7–11.2
|
7.4
|
|---|
|
c) Miscellaneous-say
|
—
|
0.9
|
|---|
|
|
|
Total 18.0
|
|---|
Elkinton and Danowski in The Body Fluids*
[ * Ref. 12-8, p. 494. ]
list data, which the writer has rounded out, condensed, and used as a basis for Table 24. These reflect the range of daily input and output for an average male weighing 155 pounds.
In medical circles it has long been believed that too much "salt" was conducive to a cardiovascular condition, and that if such condition was evident, the diet should include much less salt; hence the so called "low salt" or "salt–free" diets. From the standpoint of Physical Chemistry, the writer could not agree more. It is perhaps a minor point, but the medical profession singles out sodium chloride, (as contrasted with potassium chloride or potassium sulphate) as being the offender. On this, the writer cannot agree.
Figures 19,
Fig. 20, and Figs. 157 and 158 (pp. 232 and 233) show that any electrolyte, regardless of type, will "salt out" substantially any colloid system if carried to extreme. Therefore, in the writer's opinion, excessive potassium chloride or sodium sulphate or potassium sulphate would if present be substantially as harmful as sodium chloride. There would be some difference in the three, but regardless of such difference, all could (and would) be lethal if carried to extreme. But, as to the preference between sodium and potassium, the writer has good evidence and agrees with others who hold that potassium salts are much more favorably received by the system than sodium.
The average concentration of mineral salts in blood was given in Tables
No. 13 and
No. 14. The writer believes these values should be referred to as "average for the United States" — or "average" for the particular group which it represented. The writer cannot believe that they obtain on a worldwide basis.
It should be stated that perhaps the only reason we employ sodium chloride for "table salt" is that it is found in large quantities in the United States (and many other areas), and it is about the cheapest "food seasoning" to be found.
In Ethiopia in 1943, there were two varieties of "native salt" neither of which remotely resembled the sodium chloride we employ for human consumption. One was the residue from evaporated Red Sea water, produced commercially at Aden; the other was the residue from natural evaporation of the Awash River as it flowed Eastward toward the sea.**
[ ** The entire River "disappears" on the desert floor (due to evaporation) and forms a salt flat. When the River flows off the escarpments at "normal humidity," it drops to an elevation 5 ft. above sea level, where humidity is only 5 to 15%. Natives gather the dried plates of "salt," and barter them to desert people in North Africa. This residue was comparable in mineral composition to that from a typical limestone water found, say, in New Jersey. Is this salt (or perhaps lack of salt) associated with the fact that Yemenites, with a history of freedom from cardiovascular disease, suddenly begin to acquire it when they move into the metropolitan cities in Israel? ]
We list the composition of these two salts in
Table No. 25, together with the composition of human plasma.
|
TABLE NO. 25
|
|---|
|
Constituents
|
Typical
Ocean Water
|
Water from the
Awash River
(approximate)
|
Human
Plasma
|
|---|
|
( Column 1 )
|
( 2 )
|
( 3 )
|
( 4 )
|
( 5 )
|
( 6 )
|
|---|
|
Atomic Formula
|
mg / l ppm
|
%
|
mg / l ppm
|
%
|
%
|
|---|
Ca(HCO3)2*
*As CaCO3
|
|
|
150
|
64.0
|
|
|---|
|
MgS04
|
|
|
50
|
21.2
|
|
|---|
|
NaCl
|
23,477
|
68.3
|
5
|
2.1
|
|
|---|
|
SiO2
|
|
|
5
|
2.1
|
|
|---|
|
KNO3
|
|
|
5
|
7.1
|
|
|---|
|
MgCl2
|
4,981
|
14.4
|
|
|
|
|---|
|
Na2SO4
|
3,917
|
11.4
|
20
|
8.5
|
|
|---|
|
CaCl2
|
1,102
|
3.2
|
|
|
|
|---|
|
KCl
|
664
|
1.9
|
|
|
|
|---|
|
NaHCO3
|
192
|
0.5
|
|
|
|
|---|
|
KBr
|
96
|
0.3
|
|
|
|
|---|
|
H3BO4
|
26
|
—
|
|---|
|
SrCl2
|
24
|
—
|
|---|
|
NaF
|
3
|
—
|
|---|
|
Total
|
34,482
|
100.0
|
235
|
100.0
|
|
|---|
|
|
|---|
|
Na
|
—
|
30.5
|
|
|
41.3
|
|---|
|
Mg
|
—
|
3.7
|
|
|
0.2
|
|---|
|
Ca
|
—
|
1.15
|
|
|
1.3
|
|---|
|
K
|
—
|
1.1
|
|
|
2.1
|
|---|
|
Sr
|
—
|
0.04
|
|
|
—
|
|---|
|
Cl
|
—
|
55.2
|
|
|
46.5
|
|---|
|
S04
|
—
|
7.7
|
|
|
0.2
|
|---|
|
Br
|
—
|
0.19
|
|
|
—
|
|---|
|
HB03
|
—
|
0.07
|
|
|
—
|
|---|
|
HCO3
|
—
|
0.35
|
|
|
7.9
|
|---|
|
P04
|
—
|
—
|
|
|
0.5
|
|---|
|
Total
|
|
100.0
|
|
|
100.0
|
|---|
Note the difference in percent mineral constituents in columns 3 and 5. It is hard to believe that the blood electrolytes of three persons would be closely comparable if, for culinary and dietary purposes, the first employed only sodium chloride; the second, only ocean water residue; and the third, only residue from the Awash River. Neither are the fertilizers employed for cultivating foodstuffs the same the world over, nor the soils, which produce the foodstuffs. Can anyone today state that our mineral input is optimum in concentration and/or composition? It is pertinent to inquire "Just what is optimum?" The correct answer would undoubtedly be helpful.
The content of dissociated mineral salts of human urine will principally reflect the daily input of foodstuffs and liquids. It will vary from day to day (actually, from hour to hour) in accordance with the nature and weight of the mineral input, assuming of course that the minerals are soluble.*
[ * Relatively insoluble materials such as powdered calcium and magnesium carbonate will pass into the bloodstream only if they are solubilized by the HCl of the gastric juice. ]
We now divert the reader's attention again to
Fig. 19. As we have before noted, "normal" human blood (freshly spun–out serum) has a
Specific Conductance approximating 12,000 micromhos, equivalent to 6,000 ppm of NaCl.
Although a SC of 6,000 ppm of NaCl represents a ZP of about –18 mv, a system could contain as much as 15,000 ppm of sodium sulphate before ZP lowered to –18 mv, or 32,000 ppm of tetrasodium pyrophosphate. Thus, if the stability of the system was critical, a shift from 1:1 to a 1:2, or 1:3 electrolyte would be tremendously helpful in preventing coagulation.
Reference to
Fig. 19 also shows that although 6,000 ppm of a 1:1 electrolyte (such as NaCl) would produce a ZP of –18 mv, this same concentration of a 1:2 electrolyte (such as Na2SO4) would produce a ZP of –30 mv; and a similar concentration of a 1:4 electrolyte (such as Na4P2O7) would produce a ZP of –47 mv. Thus if one wishes to render a colloid system more disperse, this can be done by either or a combination of two ways, i.e.
a.) reduce the concentration of the electrolyte; and/or,
b.) maintain the same ionic concentration, but substitute an electrolyte having an anion of higher valence.
That is to say, substitute a 1:2 type for a 1:1; or a 1:3 for a 1:2. Thus, if our difficulty in intravascular coagulation lies in the simple condition of the blood being "salted out" by too much 1:1 type electrolyte (plus the presence of a low concentration of a 3:1 type), an answer would be to decrease the 1:1. electrolyte, and eliminate the 3:1 type. This can be accomplished in the human system by reducing our mineral intake (employing a low–salt diet); and/or by drinking an increased amount of water containing a 1:2 or 1:3 electrolyte. Whereas anionic electrolytes are by nature dispersing, most municipal water supplies are by nature "coagulating." **
[ ** Most natural waters in the United States principally contain Ca(HCO3)2 and MgS04. The former is a 2:1 and the latter a 2:2 electrolyte. Compared with 1:1 or 1:2 or 1:3 electrolytes, such waters are coagulating (rather than dispersing) in their effect. Moreover, lime is employed extensively as a primary and secondary alkali in water treatment plants. The writer does not believe that any calcium compounds should ever be employed at any municipal waterworks, except for water softening, existing concepts notwithstanding.
In the April 23, 1960, issue of the Journal of the American Medical Association (Vol. 172, pp. 1902–1908), Henry M. Schroeder, M.D., of West Brattleboro, Vermont, published an article entitled "Relation Between Mortality From Cardiovascular Disease and Treated Water Supplies." This was a rather abstract correlation of statistics, which indicated that the incidence of cardiovascular disease lowers as the hardness of municipal water increases. Schroeder closed with the statement that the data offered a "clue" to an influence of environment on cardiovascular disease. Some have interpreted the paper as an approval of calcium carbonate (which causes hardness) in water supplies. Although the writer has the very highest regard for Dr. Schroeder's work, he cannot agree with this thesis. The water supply for Roseto, Penna., the town with the lowest incidence of cardiovascular disease in the United States, is the softest natural water in the U.S.A. (This matter will be discussed in detail in Volume II.) ]
Suitable electrolytes which are dispersing in character include: sodium and/or potassium sulphate, sodium and/or potassium citrate, and sodium hexametaphosphate. One should be cautious in any attempt to employ potassium hexametaphosphate, because its high molecular weight and long–chain length could tend to induce agglomeration through mechanical bridging, if it should get into the bloodstream at low concentrations.
With regard to the size of molecules that will pass from the stomach into the bloodstream, heparin, with a molecular weight approximating 17,000, is not absorbed. This can mean that the molecular sieve (controlling this absorption) will not pass a polymer of this size. On the other hand, there seems to be no difficulty in absorbing molecules with a MW in the 1,000 to 2,000 range.
These basic principles of Zeta Potential, now well established in industrial systems, point a way to effecting much needed relief of human intravascular coagulation, and control of the Zeta Potential of blood systems.
* * * * * * * *
Again returning to the subject of blood electrolytes, although human blood the world over tends to be constant in its physicochemical characteristics, the writer firmly believes that there must be variations reflecting one's intake of foodstuffs and liquids — and their attendant mineral salts. These salts are, of course, very definitely reflected in the mineral constituents of urine.
One will note from
Table No. 24 that about half our water intake derives from food. The other liter represents intake of water per se. Thus, to double urine output requires only that we increase water input by about 1 liter. On the water output side, note that about one–half largely represents breath moisture, which remains constant. Thus, 1 liter per day of increased water intake results in 1 liter per day of increased urine output — which halves the dissolved mineral solids concentration of the urine, and reduces the "percent increase" of electrolytes from 150% to 25%.
There are many hypotheses to account for the modus operandi through which the kidney can convert blood electrolytes at 12,000 micromhos, to urine at 24,000, 30,000 or even 36,000 micromhos. These values represent concentration factors of 1:2, 1:2½ and 1:3, respectively. Although the large surface area of the kidney undoubtedly plays a prominent role in the concentration of urine, present concepts may be greatly in error.
We spent a day discussing this matter with Drs. Wayne MacRae and Daniel Brown of the Research Laboratories of Ionics, Inc., Cambridge, Mass., who specialize in the. electrochemical aspects of ion transfer. They felt that when unexplained ionic concentration (rather than dilution) was encountered, one generally looked for high temperature, high pressure, or an electrical gradient. Since there can be no high pressure or temperature involved, it suggests that the electrical impulse produced with each heartbeat (through inverse electroendosmosis) could be the driving force. According to Drs. MacRae and Brown, the significant criterion is that 26.8 ampere hours per equivalent molecular weight are required for concentration.
The principal electrolyte in blood is, of course, sodium chloride. But , when incrementally applied, all the 1:1, 1:2, 1:3 and 1:4 electrolytes (in dilute colloid systems) form a sequence. From
Fig. 19 it is evident that the Zeta Potential curve is driven to an electronegative peak; followed by a plateau; followed by a reversal of direction; then "salting out." A colloid system poised in the region of 6,000 ppm NaCl will tend to coagulate if the electrolyte concentration is materially increased, but will tend to become more disperse if the electrolyte concentration is decreased. It is also evident that a system composed of at mixture of 1:1 and 1:2, or 1:1 and 1:3 electrolytes would be more disperse than a system consisting entirely of a 1:1 electrolyte. There seems little doubt that these physicochemical relationships form the crux of the relief from intravascular coagulation.
With regard to blood electrolytes, both Selye*
[ * Refs. 11-13,11-14 and 11-15. ]
and Bajusz*
[ * Refs. 11-39, 11-40 and 11-41. ]
have stressed that they play a most important role in the overall picture of ischemia, infarction, necrosis, and cardiovascular disease in general.
It seems probable to the writer that our kidneys were not designed to operate routinely at an "overload" greater than about 25% to 50%. This would place the desirable limit of Specific Conductance of urine at 15,000 to 18,000 micromhos. Indeed it is possible they were not designed for any routine concentration. It can well be that the kidneys' ability to concentrate urine was just an emergency safeguard — comparable to an airplane operating at overload for takeoff.
The relatively high SC of urine (25,000 to 35,000 micromhos throughout most of the day and night) would strongly indicate that excess mineral salts are "stacked–up" somewhere in our system each gram awaiting its turn to be concentrated by the kidney — then passed to the bladder. The "reservoir" of temporary storage would logically seem to be our blood system. If these mineral salts concentrate in blood and appreciably raise its SC, they must tend to "salt out" the blood and thus produce intravascular coagulation.
It is also possible that through habits acquired over the past few decades (or centuries), humans drink much less water than Nature intended. It would be of interest to know the SC of the blood and urine of animals, but this evaluation is impossible since domesticated animals (or wild animals held in captivity) often eat foodstuffs (from cans) that are far removed from their native diet.
It is obvious that tests of kidney function and mineral output are best made on humans, instead of animals. This is because one can tell a human to increase his water input, and readily verify this by a check on the SC of his urine. Animals cannot be "told" to drink, and forced feeding produces "stress" — which Selye has shown produces symptoms of cardiovascular disease ranging from ischemia to necrosis.
It has long been known that the concentrations of electrolytes in the blood are controlled, probably by the glandular systems and hormones. That is to say, if one could go on a diet completely free of minerals, the blood would recycle its electrolytes, and the mineral content of the urine would tend to approach that of distilled water. However, the writer believes that the servo–mechanisms of the body will tend to keep the electrolytes of the blood at minimum, if the mineral input is low. The reverse is also true, but in a slightly different sense. Our mineral input occurs principally at meals, of which there are generally three in the first twelve–hour cycle — with none in the second. Thus after a meal, the mineral input must "stack up" in the blood system, awaiting its turn to be "pumped" to waste by the kidneys. Modern man makes available to his kidneys a very limited supply of water for dilution purposes. His kidneys, therefore, must operate almost continuously at near their maximum concentrating capacity. This is highly conducive to the maintenance of an abnormally high electrolyte concentration in the blood, which is tantamount to "salting out" the blood system.
Relief from this serious situation can be effected by significantly reducing the input of electrolytes and/or by drinking much more water — so that the kidneys can operate at a much lower concentration gradient. Beaumont demonstrated that there is no real difficulty in absorbing any reasonable amount of water from the stomach to the bloodstream, or discharging it to the bladder. I am firmly convinced that the real difficulty lies in the present need for the kidney to concentrate urine against the steep energy gradient of 1:2 or 1:3, against which it must now operate almost continuously. I do not wish to belabor this point of kidney function, but from the standpoint of Physical Chemistry, I must stress that these high concentration gradients, plus the presence of 3:1 electrolytes in our foodstuffs, appear to be the real crux of intravascular coagulation and cardiovascular disease.
It is highly germane to kidney function that human blood (serum) has a Specific Conductance approximating 12,000 micromhos — but that the SC of human urine in the United States (at present) principally ranges from 25,000 to 35,000 micromhos. This necessitates a concentration factor of 100 to 200 percent. It is amazing that the kidneys can perform against this "uphill" gradient, and it was a source of wonder to August Krogh and Homer Smith, who devoted much study to this subject.
Literature is sparse on the Specific Conductance of urine. Believing this information would throw much — needed light on kidney function, we permanently installed a conductivity cell to read the SC of urine direct in micromhos, and made it conveniently available to our employees and visitors. This has been in service for a year, and produces more data than we can process. It soon became apparent that the kidneys of all ages and both sexes produced urine principally in the range of 25,000 to 35,000 micromhos, with occasional values of 38,000, or slightly higher. Somewhat lower values, say 15,000 to 20,000 were occasionally found from 9:00 to 12:00 A.M. (One can of course, by simply controlling water intake, produce substantially any value, at any time he chooses.)
At this juncture, we wish to point to a most significant fact. Since the Specific Conductance of blood approximates 12,000 micromhos, the concentration factor (from blood to bladder) must be:
|
TABLE NO. 26
|
|---|
Specific
Conductance
of Blood —
in micromhos
|
Specific
Conductance
of Urine —
in micromhos
|
Concentration
Factor
Column 2/1
|
Concentration
by Kidney —
% increase in SC
|
|---|
|
(1)
|
(2)
|
(3)
|
(4)
|
|---|
|
12,000
|
12,000
|
1.0
|
0
|
|---|
|
12,000
|
15,000
|
1.25
|
25
|
|---|
|
12,000
|
18,000
|
1.50
|
50
|
|---|
|
12,000
|
25,000
|
2.08
|
108
|
|---|
|
12,000
|
30,000
|
2.50
|
150
|
|---|
|
12,000
|
36,000
|
3.0
|
200
|
|---|
It is obvious that if one gets rid of 18 grams of sodium chloride in 1 liter of urine, the SC of the urine will approximate 36,000 micromhos. This is just about our experience — give or take 10 to 20% on both the water and the electrolyte intake. A logical and obvious (but seldom employed) solution to the problem of kidney overload is to double the output, which halves the concentration. But the percent increase" (which in reality represents "work" done by the kidney) is lowered much more than half. Thus, if we are normally operating at 30,000 micromhos, doubling the urine output lowers this to 15,000, a ratio of 2:1. But the "percent increase" (or work done by the kidney in concentrating urine) is lowered from 150% to 25%, a ratio of 6:1.
A SPECULATIVE HYPOTHESIS TO ACCOUNT FOR INTRAVASCULAR
COAGULATION AND THE CLOTTING OF SHED BLOOD
The writer will now briefly review the basic facets of Zeta Potential that seem most pertinent to blood stability, thus extending our concepts set forth in
Chapter 14. One is at once confronted with the fact that this is a veritable jigsaw puzzle. But its apparent complexity is due largely to the condition that one of the major "pieces" is missing.
Actually, Zeta Potential, determinations of blood in its natural liquid state (in situ) would be of the greatest aid in ascertaining just "how" and "why" the system works. But unfortunately, there are no worthwhile ZP determinations extant.*
[ * There is a plethora of Zeta Potential determinations made in phosphate buffer solutions and the like. The writer does not consider them really indicative of the ZP of a specific system, as it exists in situ. But he will accept the prevalent ZP value of –17 mv as usable. ]
And this situation will prevail, until someone learns to arrest (without use of chemicals in vivo or in vitro), the triggering mechanism that induces blood coagulation. This is, and has long been, the missing piece of the jigsaw puzzle. Elucidation of this mechanism will require meticulous, unhurried research, with the highest order of cooperation from all participating agencies and individuals. In this connection, my experience with the Red Cross taught me much. And I shall not make this attempt again unless I definitely have such cooperation. There is no urgency from my standpoint. But one would think there would be urgency from the standpoint of 975,000** persons in this country scheduled to die this year of cardiovascular disease — unless more effective steps are taken to alleviate this condition.
[ ** All statistical references in this book to cardiovascular disease are from the brochure "Cardiovascular Diseases in the U.S.— Facts and Figures." This was published in 1965 by the American Heart Association in cooperation with the NHI and the Heart Disease Control Program of the PHSU.S. Dept. of HEW. ]
But despite the missing piece of the puzzle, the coagulation of blood does not seem awe–inspiring, ultra–complex, or truly difficult. It is difficult only to the extent that say electronics and radio were difficult prior to the vacuum tube of DeForest.
A reasonable hypothesis to account for intravascular coagulation and clotting of shed blood is:
1.) Blood is an aqueous system consisting principally of several plasma proteins; several electrolytes; and three formed elements — red cells, white cells and platelets.
2.) Of the plasma proteins (other than fibrinogen), we would judge that the effect of the globulins on the stability of the blood system is likely to be minimum. Their concentration is relatively low, and in the overall scheme of things, it would seem their basic functions lie along different lines from blood coagulation. However, it should be reemphasized that every anion and every cation in the system exerts its own influence (see Fig. 152, p. 227).
3.) This leaves
albumin and
fibrinogen,*
[ * It should be noted that from the standpoint of electrophoretic mobility, fibrinogen is spaced in the series of plasma proteins between beta and gamma globulin. ]
[ SPECIAL NOTE: — Certain brands of 25% albumin contain aluminum levels within the toxic range. ]
both of which appear to play key roles in blood stability. Under normal circumstances of blood flow, albumin (a lyophillic colloid with a MW of 69,000) probably exercises major control. But when the integrity of the organism is threatened through loss of "shed" blood, fibrinogen completely overrides albumin and produces a fluid gel in about five minutes — a rigid gel in ten. (See the sketch of a gel, Fig. 161, p. 238.)
4.) The blood system is one of "reasonably rigid" balance. As are all of Nature's systems, it is poised just slightly on the side of discrete (but definite) stability.
5.) Normally, with a young animal or human in good health, there is no evidence of intravascular coagulation. That is to say, the cells of the blood system are completely discrete.*
[ *It can be readily demonstrated with the stereoscopic microscope that in the case of whole milk or skimmed milk (3 to 5 days after milking, and with a ZP in the range of –17 to –19 mv) every colloid is discrete with no two particles joined. ]
6.) The blood electrolytes (considered simply as say sodium and potassium chloride and bicarbonate; and potassium, calcium and magnesium sulphate and phosphate) adsorb on, and control the stability of the albumin.
7.) The albumin (in turn) adsorbs on, and controls the stability of all the formed elements. It also adsorbs on, and renders significantly electronegative, the walls of the vascular system.
8.) Albumin serves as a tremendous bulwark of resistance against inadvertent and sudden coagulation by 3:1, 2:1 and 1:1 electrolytes, as was evident in Fig. 167, p. 245. The ingestion of a relatively high dosage of aluminum hydroxide does not immediately produce fatal intravascular coagulation, as will be later noted in the review of a report on aluminum by the Kettering Laboratory. But excessive 1:1 and/or 2:1, and/or 3:1 electrolytes can surely change the ZP of colloid systems, including those protected by albumin, or albumin — like proteins.
9.) Stability is "locked in place" to a high degree by
pH, to which the albumin (but not the electrolytes) is quite sensitive. (See Figs. 110, 118 and 121, pp. 150, 159 and 163.)
10.) The writer simply regards fibrinogen as an anionic polyelectrolyte. Its molecular weight of 300,000 and concentration of 2,800 ppm are appropriate for the formation of a rigid gel. Like albumin, it must also adsorb on all the formed elements and the walls of the vascular system. Its electronegative charge (combined with the electro negativity of the albumin at pH 7.4) is sufficient to impart significant mutual repulsion.
11.) The absolute concentration of calcium in human blood is relatively low, 100 mg/l. But even its equivalent CaCl2 in a system exerts a significant influence on Zeta Potential. Its coagulating effect is equal to 4200 ppm of sodium chloride (
see Fig. 19,). The role of calcium in blood stability is two–fold. It serves as a normal divalent (coagulating) cation in the system, and it enters into the thrombin–fibrinogen reaction in a manner which falls into a highly "catalytic" category.*
[ * Apparently all physicochemical processes, which are not really understood, fall into the category of either "catalysts" or "enzyme action." ]
12.) Calcium and thrombin are particularly and specifically adapted to the lowering of Zeta Potential, which results in the coagulation of blood. However, there are many cationic electrolytes and/or polyelectrolytes, which will (if injected hypodermically) also produce massive coagulation in situ.
13.) We will assume that the ZP of, the red cell in its "normal" state (in situ) approximates –17 mv (an EM of –1.31 microns/sec per v/cm*).
[ * This value is of course quite veritable for red blood cells in a phosphate buffer solution. It was established more than thirty years ago by Abramson. See Ref. 4-6. ]
Thus we have a stable, fluid system at constant temperature and
pH. Moreover, the system is continuously in motion, hence thixotropy must be an aid in maintaining the fluidity of the system.
So long as the Zeta Potential of the system remains constant, the fluidity (viscosity) of the system will also remain constant. But if the ZP of the system is progressively lowered by the introduction of cationic electrolytes or polyelectrolytes, then the stability of the system will undergo progressive changes — from simple agglomeration to fluid gel formation — and finally to a rigid gel.
We now arbitrarily establish a standard, setting forth varying "grades" and "degrees" of stability. And we ascribe to this classification what we consider "appropriate" values of Zeta Potential. The "Numerical Grade" is arbitrary, from zero to 10. The "Degree" will coincide with the agglomeration observed in the blood vessels of the eye when employing a horizontally aimed stereoscopic microscope.*
[ * Techniques for viewing and grading intravascular coagulation will be given in the next section. ]
Our Grades, Degrees, and Estimated Zeta Potential values, are:
|
TABLE NO. 27
|
|---|
|
Numerical
"Grade"
(arbitrary)
|
"Degree" of IVC
(Observed in
Sclera)
|
Probable ZP of
Red Blood Cells
(in situ)
|
|---|
|
0
|
Absent
|
–17
|
|---|
|
1
|
Slight
|
–16
|
|---|
|
2
|
Moderate
|
–15
|
|---|
|
3
|
Significant
|
–14
|
|---|
|
4
|
Heavy
|
–13
|
|---|
|
5
|
Very Heavy
|
–12
|
|---|
|
6
|
Terminal (death)
|
–11
|
|---|
|
8
|
Fluid gel (5 min.)
|
–7
|
|---|
|
10
|
Rigid gel (10 min.)
|
–7
|
|---|
As previously noted, this lowering of Zeta Potential in the human blood system can be induced by several procedures, including:
a.) An increase in the concentration of 1:1, and/or 2:1, and/or 3:1. electrolytes in the blood. ZP would be lowered by each type of electrolyte (on a molal basis) in the proportions of about 1 : 15 : 600, respectively.
b.) A discharge (or release) into the system of a relatively strong cationic polyelectrolyte.
We believe that intravascular coagulation is widely prevalent throughout the United States because of excessive electrolytes in our blood system. This is due to excessive use of a wide variety of mineral salts, including sodium chloride. It reflects lack of appropriate laws governing the processing of foodstuffs.
Another source of undesirable ions in the human system is the "peeling off" of metallic ions from cans; and from metallic vessels employed for processing foodstuffs.*
[ * Many cans are lined with a plastic coating, but this is often (if not generally) broken during the sealing process. When such cans are cleaned and filled with distilled water, there is (within a few days time) a significant rise in the SC of the water. We assume the rise in SC can be attributed to the presence of metallic ions. This condition should be investigated and corrected. ]
This particularly applies to
aluminum, a trivalent cation.
One cannot exclude as suspect, the presence in the drinking water (of some cities) of colloidal aluminum hydrate.**
[ ** I once selected ten cities with the highest incidence, and ten cities with the lowest incidence of cardiovascular disease in the United States. I then asked the general manager of one of our major waterworks to collect samples from them, and determine their free aluminum content. I suggested that he send duplicate samples to Dr. Henry Schroeder and to me, for our laboratory appraisal. Although the manager is a past president of the American Water Works Association, and is on their Board of Directors, I was unable to elicit any interest on his part. Apparently nobody wants to "rock the boat." I therefore just let the matter drop. These statistics, however, could be interesting. ]
This is from improper and inadequate flocculation of raw water. The concentration would be persistent, but not high. Nor can one exclude the "excessive" use of beverage alcohol, because it can be proved visually with the Sclerascope that 8 ounces of 90 proof spirits will induce significant intravascular coagulation (see Fig. 168, p. 247).
The foregoing causes are fully capable of producing grades of intravascular coagulation of 1 through 5 or 6. This agglomeration of cells reflects the short–range Van der Waals forces of attraction, and the earliest stage of "fluid gel formation." It also may reflect the presence of a very small amount of fibrin in the system–sufficient to act as a long chain polymer and bind cell to cell.
But even if these forces were combined, they are not sufficient to produce Grade 8 or Grade 10. These higher grades result solely from the release into the system of a cationic polyelectrolyte. There is the greatest probability that this polyelectrolyte exists in (or on) the platelet, and that it is simply "expressed" into the system "when the need arises." The writer has been able to prove the existence of this polyelectrolyte (by appropriate Zeta Potential techniques) for the past nine years. It is, of course, the thrombin and/or prothrombin — so thoroughly investigated by Walter Seegers. It has been known, and named and renamed dozens of times by hundreds of investigators in the medical field.
In 1952, Albritton listed five speculative hypotheses (these are repeated in Ref. 11-50) concerning the mechanism of blood coagulation. Among existing medical concepts are:
a.) The platelets are formed in the bone marrow in large cells — megakaryocytes.
b.) In due time the megakaryocytes rupture, releasing the platelets into the bloodstream where their concentration approximates 250,000 per cubic millimeter.
c.) Upon the release of blood from the vascular system to an exterior surface, the platelets (in some manner) rupture, and release some type of reagent to the system. This "contact factor" is considered to be the "triggering" mechanism.
d.) The released reagent converts thromboplastinogen (an inactive precursor in the system) to thromboplastin, a protein enzyme.
e.) The thromboplastin then converts the prothrombin molecule to thrombin, another protein enzyme.
f.) The thrombin (in the presence of an appropriate amount of calcium) then combines with the fibrinogen (another inactive precursor) to form fibrin — the polymer Which enmeshes the water of the blood and forms a rigid gel.
The writer sees no need for any "enzyme action" unless the change in Zeta Potential, due to the platelet discharging a cationic polyelectrolyte to the system, can be so termed. From the writer's point of view, one only clutters the picture with concepts of thromboplastinogen, thromboplastin, prothrombin, and enzyme action. The simple concept of Zeta Potential, wherein a cationic polyelectrolyte is contained in and released by the platelet to the system, is ample to account for the gelation noted.
Neither can the writer consider that heparin is connected with or related to enzyme action — existing concepts notwithstanding. If we consider that heparin involves enzyme action, then we must also consider that laundry detergents are effective because of "enzyme action."
Another highly pertinent but unanswered question in the field of blood stability is: At the end of the 8 day period of the platelet's useful life, how is it "defused"? Does the cationic polyelectrolyte gradually deteriorate and lose its electropositive characteristics? Does it remain active, and is it revitalized in the next "batch" of platelets? Is it neutralized by an anionic electrolyte or polyelectrolyte? Or what?
One more pertinent and unanswered question: "Does the platelet ever undergo a slow leak into the system?" And is this a major cause of intravascular coagulation and cardiovascular disease?
From the writer's point of view, it is pointless to pose these questions seriously until we first answer the major question of: What is the nature of the triggering mechanism which causes the platelet to discharge its polyelectrolyte — and only at a specific, selected, and appropriate time and location? The writer does not know the answer, but he does have one physicochemical principle, which has not been tried that conceivably could be the answer. Or it can well be that the answer will never be known. The writer will not outline this principle because someone inexperienced in Zeta Potential might try it out — fail — and then conclude that the principle did not apply. This has happened before. The writer does not believe that this can be a contact factor. The surfaces involved are just too small. On Figs.
170 and
171, we show human blood platelets. This electron micrograph is taken from Coagulation and Transfusion in Clinical Medicine, through the kind courtesy of Johnson and Greenwalt.*
[ * Ref. 11-19. ]
When time becomes available, the writer plans to work briefly with Frank van den Bosch of Cedar Grove, N. J., whose ultraviolet microscope** is now set up at Downstate Medical Center in Brooklyn.
[ ** This microscope projects an image on a fine–lined TV screen. Magnification of 6,000 to 30,OOO X is possible, and no desiccation is required. Therefore, it is well adapted to viewing and photographing highly motile microorganisms — and blood cells. ]
If we are successful in overcoming some limitations of time, dilution and tonicity, the writer believes it is possible we may be able to actually see the platelet exude its liquid to the system.
In bringing this section to a close, it should be noted that the writer's simple hypothesis of blood agglomeration and coagulation is in agreement with all the precepts of Zeta Potential thus far developed for industrial colloid systems. In relating these principles to blood, it was not necessary to introduce any new facets to make blood stability conform to the overall pattern of industrial colloid stability — as controlled by Zeta Potential. And as far as we know, there is nothing in this hypothesis that seriously conflicts with the dozen–odd "factors" which represent the medical approach to blood coagulation.
EVALUATION OF INTRAVASCULAR COAGULATION WITH THE HORIZONTALLY AIMED STEREOSCOPIC MICROSCOPE
After designing the Sclerascope,*
[ * This instrument is available from Zeta–Meter, Inc., on special order. ]
an adjustable "stock" for holding the head steady, it was readily possible to observe the presence or absence of agglomerates in the blood vessels situated in the white of the eye. We soon had no difficulty in distinguishing between a low and a high degree of agglomeration. But what we required was a graduated scale to denote progressively increasing degrees of Intravascular Coagulation (IVC). This necessitated the preparation of definitions and "ground rules" covering the semantics of colloid stability. *
[ * These were included in Chapter 20. ]
We attempted to have these express what is seen with the microscope, and also convey some concept as to the origin of the forces that brought and held the particles together.
We gradually built up a system of classification, numerically ranging from Grade zero to Grade 6. The former represented no detectable agglomeration or its symptoms; the latter, tremendous agglomeration — resulting in significant sedimentation in horizontally positioned large diameter vessels.
Details of the Sclerascope are shown on
Fig. 172. Illumination of the left eye *
[ * Only one eye is examined. However, if necessary, the position of the microscope may be changed to view the right eye. ]
is provided by a thin beam of blue–white light, projected almost at right angles to the line of vision. Light intensity is variable, and heat is removed by a filter. The "head stock" is adjustable in both a vertical and left–right traverse. The stereoscopic microscope is positioned horizontally, and is adjustable in both vertical and horizontal planes.
The face of the person undergoing examination is held steady and in a frontal position by the headstock. The eyes are turned about 15° upward and 45° to the right, focusing on a target spot. This enables the conjunctiva of the left eye (as far as the cornea) to be readily illuminated, and minimizes glare, eyestrain and fatigue.
In selecting an optical system, one might at first assume that the higher the overall magnifying power — the better the view of the blood vessel. This is not the case. Available with American Optical Company's equipment are six "fixed" objectives (1, 2, 3, 4, 6 & 8X), and two eyepieces (15X & 2OX). As the magnification of the objective is increased, the "depth of sharp focus" becomes thinner. This situation is worsened by the natural curvature of the eye, and inability of the person under study to remain motionless during the examination. These considerations rule out use of the 6X and 8X objectives, leaving only the 1, 2, 3 and 4X. Since the nosepiece of the microscope will accommodate only three objectives, one of these must be excluded.
The writer has worked for long periods with 1–2–3X, and 2–3–4X, and 1–3–4X objectives, using both 15 and 20X eyepieces. The 1–2–3X combination with 20X eyepieces, has proved the most satisfactory. This of course gives overall magnifications of 20, 40 and 6OX. These considerations are of prime importance because proper evaluation of the "degree of intravascular coagulation" depends, to a large extent, upon how clearly and definitively one can see the flow of blood in the arterioles and venules. It should be stressed that the microscope must be carefully aligned, particularly with regard to collimation.
In viewing the "white" of the eye, one first sees the bulbar conjunctiva — the thin, opaque mucous membrane covering the anterior portion of the globe of the eye. This membrane is heavily laced with blood vessels, which are readily viewed with the microscope. The sclera, the firm fibrous outer layer of the eyeball, is covered by the conjunctiva. It is likewise heavily laced with visible blood vessels, but because of their position (to the rear) they do not stand out as plainly as those in the conjunctiva. Hence the flow of blood in the sclera is somewhat less visible than in the conjunctiva.
The 1X objective is employed in scanning the conjunctiva and sclera to locate the area containing the "critical" vessel or vessels. We term these vessels (and their surrounding areas) critical because it is here that the physical manifestations of intravascular coagulation are most clearly revealed, and judgment is made. However, in severe cases, substantially the entire conjunctiva and sclera give evidence of intravascular coagulation. The location of these critical vessels will vary from eye to eye, because the vascular pattern of each individual differs — to a far greater extent than the thumbprint. It should be stressed that evaluation is generally made on the basis of a "detail" examination of very few arterioles or venules; and often only one. Regardless of the relative degree of coagulation from day to day or week to week, maximum coagulation will generally be found (in any given individual) in the same venules or arterioles. This is due to hydraulic considerations, because these vessels will have optimum diameter, position, configuration and vascular connections to maximize the manifestations of coagulation.
After selecting the critical vessel (or vessels) with the 1X objective, confirmation and further study is made with the 2X objective. The 3X objective is then employed for obtaining an enlarged and more detailed view of the vessels under examination. An overall magnification of 6OX is necessary for this final evaluation. Consequently ophthalmic microscopes having a maximum magnification of 3OX are generally unsuitable.
One should examine both the conjunctiva and the sclera. The blood vessels of the conjunctiva stand out clearer and are therefore more easily studied. One will note, however, that "clumping" is often more pronounced in the sclera than in the conjunctiva. This is because frequent blinking of the eye during an examination tends to mechanically break up agglomerates and clumped cells in the conjunctiva. The vessels of the sclera are not affected by blinking.
Appropriate examinations of the conjunctiva and sclera permit evaluations of progressive stages of intravascular coagulation, which are well in advance of cardiovascular emergency episodes. We have classified seven degrees of intravascular coagulation, which we list herein, with a definition of terms:
COMETS
A "Comet" results from a capillary being filled principally with clear plasma, instead of being filled uniformly with erythrocytes and other blood cells. Red cells will then "dart" singly or in small groups through the capillary, giving the impression of "shooting stars" or comets. The linear spacing from blood cell to blood cell often exceeds five to ten times the diameter of the capillary. At times, the capillary may contain only one or two "darting" cells. In health, most of the capillaries in service*
[ * Krogh showed that under normal conditions of sedentary work or inactivity, only a small percentage of capillaries (say 10 to 15) are in actual service. The percentage of capillaries in service varies directly with physical exercise and oxygen requirements. When a person is undergoing maximum physical exertion, all capillaries are in service. ]
are substantially filled with discrete cells, and the flow is continuous and uniform. Comets are perhaps the first indication of intravascular coagulation. It would seem that the reason comets exist is that if every blood cell is free and discrete, with no aggloiheration, its distribution in the overall system will be uniform; but if some cells are agglomerated and some discrete, it follows that the discrete cells will have more interstitial space in which to move.**
[ ** The actual volume available for the containment of blood in arteries, artery, veins and venules is controlled to a degree by vasoconstrictors and vasodilators. Thus blood pressure, in health, is maintained fairly constant (and blood vessels remain filled) regardless of blood volume. ]
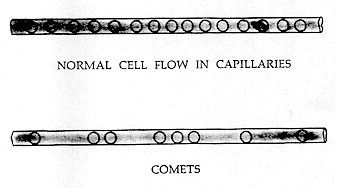 The onset of intravascular coagulation is therefore characterized by a few darting cells in a few capillaries. An increased degree of coagulation is manifested by a greater number of darting cells in a greater number of capillaries. It should be noted that Comets characterize only the first and second degrees of coagulation, becoming less noticeable at higher degrees.
The onset of intravascular coagulation is therefore characterized by a few darting cells in a few capillaries. An increased degree of coagulation is manifested by a greater number of darting cells in a greater number of capillaries. It should be noted that Comets characterize only the first and second degrees of coagulation, becoming less noticeable at higher degrees.
This state is shown on Fig. 173. (left)
AGGLOMERATION
Agglomeration is the condition when blood has partially lost its "fluidity," and cells flow in rope or chain–like fashion as though one cell was joined to another. (This is not to be confused with rouleau formation.) The normal "turbulent" flow appears to change, as the individual cells lose their "discrete" mobility. This is undoubtedly brought about by a lowering of Zeta Potential, which results in the formed elements becoming individually enmeshed in the first stages of a "fluid gel." One may also hypothesize that discrete strands of fibrin have formed to sufficient length to encapsulate the red cells and loosely bind one cell to another. This "cohesiveness" imparts a "rope–like" tendency to the flow pattern, with each cell closely following the path of the preceding one. One might classify the physical appearance of this blood flow as "coarsely granular." This "rope–like" flow is in sharp contrast to "turbulent flow" — wherein each cell stands out boldly and seems to change its position continually in the general flow pattern.
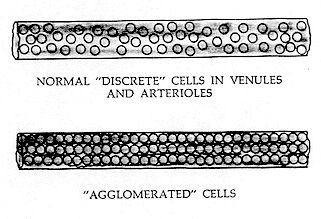 Agglomeration is best evaluated in arterioles or venules, which are 30 to 60 microns in diameter, or 4 to 8 times the size of a capillary. Because of opacity, discrete cells cannot be readily seen in the largest veins or arteries of the eye; and because they move too rapidly, they cannot be satisfactorily observed in the capillaries.
Agglomeration is best evaluated in arterioles or venules, which are 30 to 60 microns in diameter, or 4 to 8 times the size of a capillary. Because of opacity, discrete cells cannot be readily seen in the largest veins or arteries of the eye; and because they move too rapidly, they cannot be satisfactorily observed in the capillaries.
STASIS
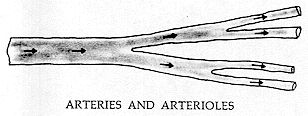 Arteries and arterioles can be distinguished from veins and venules in the sclera and conjunctiva. Arteries branch and flow to the (smaller) arterioles, single file only. With clumping, each cluster of cells which in turn branch and flow to the (smaller) capillaries. Thus, the direction of flow follows the branching.
Arteries and arterioles can be distinguished from veins and venules in the sclera and conjunctiva. Arteries branch and flow to the (smaller) arterioles, single file only. With clumping, each cluster of cells which in turn branch and flow to the (smaller) capillaries. Thus, the direction of flow follows the branching.
 Veins and venules show the opposite pattern. Capillaries flow to the (larger) venules; venules to the (larger) veins. The direction of flow is, therefore, counter to the direction of branching.
Veins and venules show the opposite pattern. Capillaries flow to the (larger) venules; venules to the (larger) veins. The direction of flow is, therefore, counter to the direction of branching.
Partial stasis is when the rate of blood flow is markedly reduced in arteries, arterioles, veins or venules. A higher degree of stasis is evident when the flow periodically comes to a faltering halt — then actually reverses for a few seconds — then sluggishly flows again in its normal direction. In advanced stasis, a mass of cells may traverse backward and forward in a venule or arteriole for as long as five to thirty seconds before resuming normal flow. Stasis, in its varying degrees, is an important criterion in judging intravascular coagulation.
In health, and in the absence of intravascular coagulation, blood flow is brisk, uniform and unfaltering — from arteries to arterioles to capillaries to venules to veins. There is no significant stasis or even temporary reversal of flow.
CLUMPING
The term "clumping" is used when cells adhere to each other, forming individual groups of about 5 to 100. In reality, this is a worsening of the condition previously referred to as "agglomeration." Clumps may be found in arteries, veins, arterioles and venules. They are not evident in capillaries, where the small diameter permits red cells to traverse in single file only.
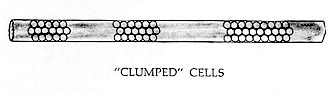 With clumping, each cluster of cells is separated from its adjacent group by a "cylinder" of clear plasma. The length of these clumps is generally two to five times the diameter of the venule or arteriole. Conversely, the length of the clear cylinder is more often one to two times the diameter of the lumen, as shown in the drawing.
With clumping, each cluster of cells is separated from its adjacent group by a "cylinder" of clear plasma. The length of these clumps is generally two to five times the diameter of the venule or arteriole. Conversely, the length of the clear cylinder is more often one to two times the diameter of the lumen, as shown in the drawing.
SEDIMENTATION OF CELLS
This condition, which is sketched below, has been thoroughly covered in pictures and text by Knisely and his associates — and we refer the reader to their published works. (Ref. 11-16.)
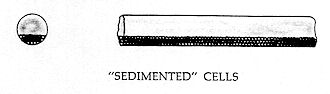
Based on the foregoing definitions and illustrations, the writer categorizes the seven degrees of intravascular coagulation as:
|
Grade
|
Characteristics
|
|---|
|
0
|
No comets, agglomeration, stasis, or clumps.
|
|---|
|
1
|
Some comets.
|
|---|
|
2
|
Many comets. Noticeable chain–like agglomeration of blood cells.
|
|---|
|
3
|
Pronounced chain–like agglomeration. Some stasis. Some clumping.
|
|---|
|
4
|
Noticeable stasis and noticeable clumping in some blood vessels.
|
|---|
|
5
|
Pronounced stasis in some blood vessels. Pronounced clumping in many blood vessels.
|
|---|
|
6
|
Pronounced stasis and heavy clumping in many blood vessels. Settlement of cells in a few horizontally–positioned veins and/or arteries, the red
cells occupying the lower quarter to half of the lumen, with plasma containing the white cells seen above. This represents Knisely's Stage III — after which all 75 of his Rhesus monkeys died within 3 to 12 hours.
|
|---|
These characteristics are set forth in Table No. 28.
|
TABLE NO. 28
|
|---|
|
GRADES AND DEGREES OF INTRAVASCULAR COAGULATION
|
|---|
|
Grade
|
Degree of IVC
|
Comets
|
Chain–like Agglomeration
|
Partial Stasis
|
Clumping
|
Sedimentation in lumen
|
|---|
|
0
|
Absent
|
Absent
|
Absent
|
Absent
|
Absent
|
Absent
|
|---|
|
1
|
Slight
|
Some
|
Absent
|
Absent
|
Absent
|
Absent
|
|---|
|
2
|
Moderate
|
Many
|
Noticeable
|
Absent
|
Absent
|
Absent
|
|---|
|
3
|
Significant
|
—
|
Pronounced
|
Some
|
Some
|
Absent
|
|---|
|
4
|
Heavy
|
—
|
—
|
Noticeable in some vessels
|
Noticeable in some vessels
|
Absent
|
|---|
|
5
|
Very Heavy
|
—
|
—
|
Pronounced in some vessels
|
Pronounced in many vessels
|
Absent
|
|---|
|
6
|
Terminal
|
—
|
—
|
Pronounced in many vessels
|
Heavy in many vessels
|
Noticeable in a few vessels
|
|---|
Forty to fifty percent of the persons in the age bracket of 25 to 65 who were examined by the writer have shown Grade 3 or higher. One male (age 75) with Grade 5 coagulation, died shortly after being forced to climb two flights of stairs during the New York City electric power blackout (November 1965). Another male (age 63) with a known but inoperable tumor of the pancreas was classified Grade 5. He died of cancer two months later.
The examination of the sclera and bulbar conjunctiva with the horizontally–aimed microscope is highly essential to the evaluation of intravascular coagulation. There is every evidence that IVC is closely allied to the physicochemical manifestation of cardiovascular disease.
Minor illness (such as head colds or "two–day virus") will, in 24 hours, increase an IVC grade of 0–1 to Grade 3, and occasionally to Grade 4. It is probable that substantially all persons who are gravely ill with a disease caused by a microorganism will show a degree of Grade 4 to Grade 5. Indications are that there is some increase in IVC during the menstrual period; and that high blood pressure (through vasoconstriction) has a tendency to mask intravascular coagulation.
EXAMPLES OF INTRAVASCULAR COAGULATION BY MELVIN KNISELY
We now show several pages of highly pertinent Figures from various technical papers by Melvin Knisely and associates (Refs. 11-16-b, d, e, and i). We particularly recommend Ref. 11-16-i, which contains an excellent summary of his work and many valuable sources of reference material.
We are confident that Knisely's work will go down in history as one of the major contributions of this generation to biology and physiology.
 The gravitational separation of plasma, white cells and red cells. Note that some of the white cells are in clumps.
The gravitational separation of plasma, white cells and red cells. Note that some of the white cells are in clumps.
 Diagram showing condition of the blood in the inferior vena cava of the monkey whose blood had been changed to a sludge by Knowlesi malaria. The vessel was between ½ and ¾ inch in diameter. 1. The lowest one–fourth to one–third of the vessel is covered with a paste made up of settled, stationary masses of agglutinated parasitized and unparasitized red cells effectively segregated from the "circulating blood". 2. The middle third consists of somewhat smaller masses carried slowly forward in suspension. 3. The top third consists of supernatant plasma, containing a few small suspended masses all moving forward more rapidly. The flow, if it can be called "flow", of the moving materials was from right to left.
Diagram showing condition of the blood in the inferior vena cava of the monkey whose blood had been changed to a sludge by Knowlesi malaria. The vessel was between ½ and ¾ inch in diameter. 1. The lowest one–fourth to one–third of the vessel is covered with a paste made up of settled, stationary masses of agglutinated parasitized and unparasitized red cells effectively segregated from the "circulating blood". 2. The middle third consists of somewhat smaller masses carried slowly forward in suspension. 3. The top third consists of supernatant plasma, containing a few small suspended masses all moving forward more rapidly. The flow, if it can be called "flow", of the moving materials was from right to left.
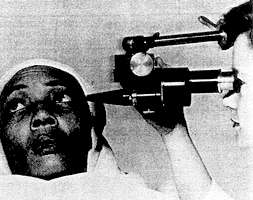
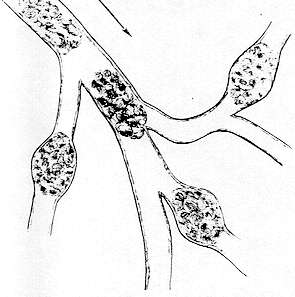 Diagram of rather rigid, elastic masses of agglutinated red cells coming down a stem arteriole and passing out through terminal arterioles. At the upper left, the wall of the stem arteriole resists distention sufficiently so that as each mass passes through it, the mass is forced to distort toward fitting the internal form of the artery. As each mass passes down into the terminal arteriole, it arrives at a point at which the force exerted by the thin–walled terminal arterioze is considerably less than the force exerted by the thicker–walled stem arteriole. Each mass then rounds up by means of its own internal elasticity and approaches a spherical form. Note the three masses farthest out in the portrayed vessels. It is not at all uncommon to observe masses going through these reactions as they pass through the terminal arterioles and pre–capillary arterioles of the bulbar conjunctiva of diseased humans. The significant point here is that the masses have internal elasticity and return toward a spherical form as they pass into the soft–walled vessels.
Diagram of rather rigid, elastic masses of agglutinated red cells coming down a stem arteriole and passing out through terminal arterioles. At the upper left, the wall of the stem arteriole resists distention sufficiently so that as each mass passes through it, the mass is forced to distort toward fitting the internal form of the artery. As each mass passes down into the terminal arteriole, it arrives at a point at which the force exerted by the thin–walled terminal arterioze is considerably less than the force exerted by the thicker–walled stem arteriole. Each mass then rounds up by means of its own internal elasticity and approaches a spherical form. Note the three masses farthest out in the portrayed vessels. It is not at all uncommon to observe masses going through these reactions as they pass through the terminal arterioles and pre–capillary arterioles of the bulbar conjunctiva of diseased humans. The significant point here is that the masses have internal elasticity and return toward a spherical form as they pass into the soft–walled vessels.
 This vessel contained slowly moving settled masses. Note (1) large masses which completely fill the lumen of the vessel, (2) small masses settled between large ones, and (3) that the settled sludge in the upper right portion fell onto the lower internal surface of the vein as it came down around the vertically–placed bend.
This vessel contained slowly moving settled masses. Note (1) large masses which completely fill the lumen of the vessel, (2) small masses settled between large ones, and (3) that the settled sludge in the upper right portion fell onto the lower internal surface of the vein as it came down around the vertically–placed bend.
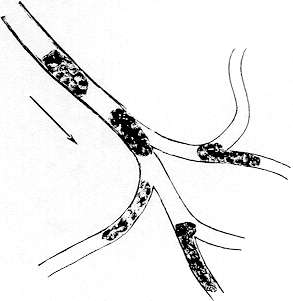 Diagram of soft plastic masses passing down an arteriole into terminal arterioles. Note that these masses, unlike those shown in photo to the left, do not round up again as they pass into the narrow soft–walled vessels. These masses are undergoing a plastic deformation.
Diagram of soft plastic masses passing down an arteriole into terminal arterioles. Note that these masses, unlike those shown in photo to the left, do not round up again as they pass into the narrow soft–walled vessels. These masses are undergoing a plastic deformation.
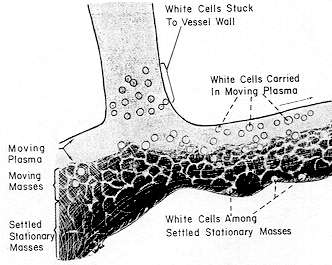 Cinetracing of a small vein containing. white blood cells in 3 separate types of location; stuck to the wall of the vessel, as described by E. R. and E. L. Clark, entrapped between large masses of settled, stationary erythrocytes located on the bottom of the vessel, and carried freely suspended in the supernatant horizontally moving plasma.
Cinetracing of a small vein containing. white blood cells in 3 separate types of location; stuck to the wall of the vessel, as described by E. R. and E. L. Clark, entrapped between large masses of settled, stationary erythrocytes located on the bottom of the vessel, and carried freely suspended in the supernatant horizontally moving plasma.
 Gravitational separation of settled red–cell masses from suspended circulating white cells. One significant consequence is that any white cell carried along in the flowing supernatant plasma cannot come in contact with, hence cannot ingest, a bacterium deep within the settled stationary blood–cell masses (cinetracing).
Gravitational separation of settled red–cell masses from suspended circulating white cells. One significant consequence is that any white cell carried along in the flowing supernatant plasma cannot come in contact with, hence cannot ingest, a bacterium deep within the settled stationary blood–cell masses (cinetracing).
 A photograph taken by transmitted light through a horizontal microscope, showing some of the settling phenomena in flowing frog blood within a venule approximately 500 µ in diameter.
A photograph taken by transmitted light through a horizontal microscope, showing some of the settling phenomena in flowing frog blood within a venule approximately 500 µ in diameter.
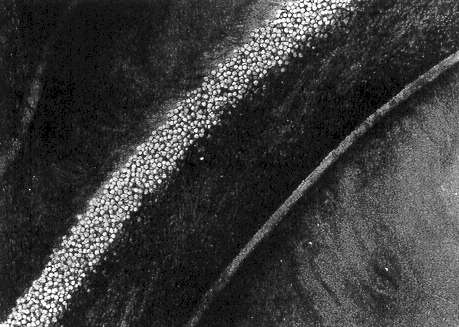 Gravitational separation of settled red cell masses from suspended circulating white cells. This is a tracing from a motion picture taken through a horizontally aimed microscope. Note the layer of concentrated white cells carried in suspension in the plasma.
Gravitational separation of settled red cell masses from suspended circulating white cells. This is a tracing from a motion picture taken through a horizontally aimed microscope. Note the layer of concentrated white cells carried in suspension in the plasma.
 Same artery as shown in
figure on page 268, seen at a later time. The flow shown here was almost quantitatively separating cell–free plasma from blood cells with just enough plasma between them to lubricate their mutually opposing surfaces and thereby permit forward motion.
Same artery as shown in
figure on page 268, seen at a later time. The flow shown here was almost quantitatively separating cell–free plasma from blood cells with just enough plasma between them to lubricate their mutually opposing surfaces and thereby permit forward motion.
Note that the capillaries of the system are partly obstructed. There were short segments of capillaries in true stasis. This is a most significant phenomenon.
THE REGIMEN
After establishing techniques and criteria for the evaluation of intravascular coagulation (IVC) as viewed in the conjunctiva and sclera, examinations of our immediate office staff (about a dozen) revealed that half of us had Grade 3 to 4 IVC; and half had less than 3. Later, we were joined by several others who had some type of advanced cardiovascular disorder. Fortunately, we were able to secure the services of several physicians, as required, in exchange for information on Zeta Potential and intravascular coagulation. They examined our group and counseled us as far as their time permitted.
Electrocardiograms with simultaneous phonocardiograms, and examinations with the Sclerascope, showed that some of our group were heavily coagulated and had arrhythmia and pre–ventricular contractions; some had heavy coagulation with no malfunctional beats; and some had moderately low intravascular coagulation (Grade 1 or 2), with no atypical beats. My two secretaries (female, age 45 and 65) had heavy IVC but no atypical beats. The writer had heavy IVC with PVC's every four to six beats, some degree of arrhythmia, and an attack of paroxysmal tachycardia every month or so. One of our group had a PVC which failed to discharge blood every other stroke; and another, a similar PVC every third stroke. We established (and maintain) what we term a "Regimen." This consists of drinking sufficient (specially prepared) water to limit Specific Conductance of the urine to a normal maximum of about 18,000 and preferably 15,000 micromhos. As predicted, this requires drinking about four to six 8–ounce glasses of the water per day, which (at 250 ml per glass) amounts to 1.0 to 1.5 liters daily. Experience has shown that eight glasses are more than required, and four are absolute minimum. This water is in addition to that routinely consumed in tea, coffee and/or milk. The composition of five types of specially prepared water with dispersing characteristics is shown in Table No. 29.
|
TABLE NO. 29
|
|---|
|
COMPOSITION OF SPECIALLY PREPARED WATER WITH DISPERSING CHARACTERISTICS
|
|---|
|
( Normal concentration for drinking — 1.0 grams per liter )
|
|---|
|
Formula Number
|
Electrolyte
|
Formula
|
MW
|
Type of Electrolyte
|
Millimols / l @ 1.0 gpl
|
|---|
|
1
|
Sodium Sulphate
|
Na2SO4 — Anh.
|
142
|
1:2
|
7.05
|
|---|
|
2
|
Potassium Sulphate
|
K2SO4 — Anh.
|
174
|
1:2
|
5.75
|
|---|
|
3
|
Sodium Citrate
|
Na3C6H507 • 2H20
|
294
|
1:3
|
3.4
|
|---|
|
4
|
Potassium Citrate
|
K3C6H5O7 • H20
|
324
|
1:3
|
3.10
|
|---|
|
5
|
Sodium Hexametaphosphate
|
14NaPO3 • Na2O
|
1490
|
1:3(Eqv.)
|
0.67
|
|---|
We have continuously employed these electrolytes, singly or in combinations, over a period of about two years. Preparation is as follows: A "Stock Solution" of 50 grams of reagent is made up to one liter with distilled water. If reagents are mixed for example, sodium citrate + potassium citrate, then the weight of the two combined is equal to 50 gpl. So far, we have tried Stock Solution combinations of sodium–potassium sulphate, sodium–potassium citrate; potassium citrate; sodium hexametaphosphate, etc., in the following ratios: 50–0; 35–15; 25–25; 15–35; and 0–50 grams per liter. We find so far that of the five reagents — and the possible combinations — 50 gpl of straight potassium citrate is the most effective. We add 10 grams of potassium (or sodium) bicarbonate to all Stock Solutions, regardless of formulation. This raises the
pH of the Standard Solution to 8.0–8.4. We consider it highly essential to maintain the pH of water somewhat above the normal pH of blood (7.4.) This ten grams of bicarbonate is employed solely for pH control, and is in addition to the 50 grams of dispersants.
From the 50 gpl Stock Solution, we prepare (as required) a Standard Solution by diluting 15, 20, 25, or 30 ml of the Stock Solution to one liter with distilled water. It is obvious that 20 ml per liter of stock solution gives a concentration of 1.0 grams per liter of the electrolyte (1,000 ppm, 1,000 mg/l). At 1.5 liters of water per day, this naturally equals 1.5 grams of electrolyte per day. Both Stock and Standard solutions should be kept in the refrigerator. None of our group is on a "low–sodium" diet of any sort, but we all attempt to control our salt consumption. The smoking and drinking pattern ranges from "moderate" to "abstinence" — upon a purely voluntary basis.
Reagent dosages for our group are always adjusted upon the basis of sclerascopic examinations and electrocardiograms. The periodicity of these examinations is geared to meet changing situations. It may vary from as few as one every two months, to bi–weekly.
Brief comments, based on our lengthy consideration of this problem, are as follows:
1.) A significant lowering of existing IVC and/or PVC* cannot be attained by drinking any amount of untreated distilled water — or tap water.
[ * IVC signifies Intravascular Coagulation; and PVC, Pre–ventricular Contraction (a malfunctional heartbeat which seldom throws blood). ]
We have no convincing evidence but it seems reasonable that if in early childhood one establishes the habit of drinking a relatively large amount of water, this should be a deterrent to IVC and PVC's in later years.
2.) Except in cases of absolute unavailability, Stock and Standard solutions should be made with distilled water — not tap water.
3.) Potassium compounds are more effective than sodium compounds in relieving PVC and IVC.
4.) The 1–3 electrolytes are considerably more effective than the 1:2. This, of course, agrees with all basic concepts of Zeta Potential.
5.) As a generalization, a dosage of 20 ml/liter approximates optimum concentration. We usually bring up the dosage slowly: 15, 17.5, 20, 22.5 and 25 gpl of "Stock Solution" per liter of distilled water, being guided by the Sclerascope and ECG.
6.) In instances where PVC's were initially found, a reduction in the Grade of IVC has always been accompanied by a reduction in the percentage of PVC'S. Where a Grade of IVC of 3+ to 4 is found, it is often difficult to lower this to a Grade less than 2. Lowering to Grade 1 is sometimes possible — but it requires meticulous adherence to the formula, and smoking and alcoholic beverages must be held to a minimum. We consider that avoidance of
aluminum is mandatory.
7.) Indications are that time is of the essence in correcting a PVC. The longer the duration and the higher the percentage of PVC'S, the more difficult is the correction. The sooner remedial steps are taken after PVC's are discovered, the simpler the task and more certain the recovery.
8.) Arrhythmia usually responds well to the regimen, and complete elimination has been obtained in a number of instances.
9.) Under our Regimen, the rate of reduction of PVC's generally forms a pattern. Usually it is a reduction of malfunctional beats from say 30 to 15 to 7 to 4 to 2 to 1% in about 2½ months. Such reductions sometimes occur in less than half this time. With very difficult situations, the pattern may level off in the 1 to 5 or even 5 to 10% range.
10.) The PVC which occurs as a "double" (two PVC's in succession) is more difficult to relieve than a single. A PVC which occurs as a triple, is much more difficult than a double. In this respect, the ratios seem comparable to the concept of the "ionic strength function" of 1:3:6.
11.) When PVC's are found in the 1 to 5% range, with no dual malfunctions, control can often be effected with 1:2 electrolytes such as potassium sulphate. More difficult situations require 1:3 (Or equivalent) electrolytes.
12.) If one abstains from the Regimen for a period of 24 to 48 hours, the Grade of the IVC and the percentage of PVC's may increase appreciably. In this respect, the Regimen is quite comparable to heparin.
13.) If a person on a 1:3 electrolyte substitutes a 1:2, PVC's may appreciably increase. Upon return to the 1:3 electrolyte, the percent malfunction is again lowered.
14.) None of our group now employs aluminum cookware of any sort, or partakes of foodstuffs that are known to have been in contact with aluminum.
15.) It seems necessary to maintain this Regimen daily, and for "life." In this respect, it is similar to heparin or eyeglasses. Thus far, in only one instance has an individual been able to discontinue the Regimen without any resumption of PVC'S.
16.) The SC of one's urine can be kept in the range of 8,000 to 16,000 micromhos until noon if one drinks 2½ glasses of the Regimen water before, during, or immediately after breakfast.
17.) We agree with those who stress the need for a low–fat, low–calorie diet.
18.) There are many appropriate anionic electrolytes and polyelectrolytes available. Some hold as much or more promise than the ones we presently employ.
19.) It is well established in medicine that because of hemorrhage, one must guard against overdosage of heparin. There has been no indication that there is such danger in any of the reagents we have employed.
20.) It should be stressed that none of our reagents could be employed in "capsule" form, because without the accompanying water, they would actually increase the "salting out" effect, and the degree of IVC. An absolute minimum of one liter (4 glasses) is required. One and one–half liters (6 glasses) is often preferable. It should be spaced throughout a 12 to 16 hour day.
21.) We believe that heparin is simply one of a great many anionic dispersing agents, which can be employed. It is readily evaluated by Zeta Potential. But we see no relevance to, or connection with, enzyme action. Neither do we believe that this "anionic polymer approach" to the control of cardiovascular disease complies as closely with "natural states" — as does the use of simple anionic electrolytes.
22.) Without doubt, the real answer to cardiovascular disease is to cease the practice of "salting out" the human blood system, and eliminate all ingestion of 3:1 electrolytes. Despite the fact that the basic chemical principles involved were common knowledge to Schulze and Hardy 87 years ago, the FDA still does not have the slightest concept of them. Their preoccupation with trivia, and inability to face real issues and hard facts, brings to mind the compulsive drive of the lemmings as they head out to sea.*
[ * See Note 3, page 335. ]
23.) Distilling water each week in the home, preparing solutions, and drinking four to six glasses a day at home or at the office — wherever you are — is something of a chore. But to the writer (and others on this Regimen) it seems preferable to having a hypodermic injection twice a day for life — or dying of heart disease.
24.) We have noted no untoward side effects from the daily ingestion of 1.5 grams of the reagents listed in
Table 29. Any of the electrolytes taken internally at a dosage of 10 to 20 grams would have a pronounced laxative effect. In fact, magnesium citrate is widely employed as a laxative. At 1.0 gram per liter, one cannot detect any taste of the reagent in distilled water.
25.) For obvious reasons, stills with block tin (not aluminum) condensers should be employed.
[ Tin can also quickly become toxic in the human system. Stainless steel or lead–free glass are the only truly safe materials that should be used. Reverse Osmosis units are now available for homes, and provide an energy efficient alternative. — TRC — ]
For the small user, distilled water is preferable to deionized. Deionizers are suitable for large, well–supervised installations, but they are not recommended for small users because of possible bacterial growths.
26.) It is not possible in one's daily life to always maintain urine at SC less than 15,000 or 18,000 micromhos. But this Regimen should be maintained as far as possible.
27.) A strict Regimen of no tobacco, no alcohol, selected foodstuffs and daily exercise is insufficient to eliminate existing intravascular coagulation. Neither can the situation be appreciably relieved by the intake of 1.5 liters/day of untreated distilled or tap water. Distilled water with an appropriate concentration of a dispersing agent is absolutely necessary.
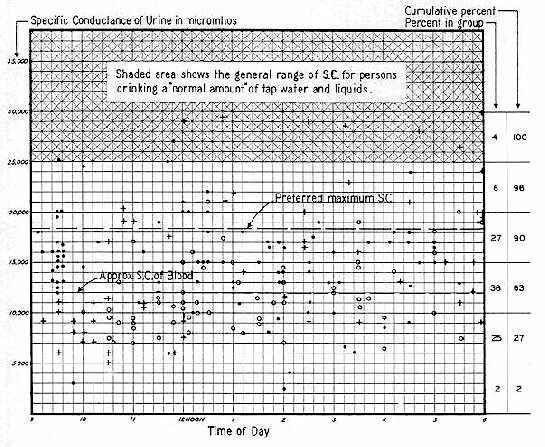
Figure 183 shows the SC of urine from several of our group over a period of a month. Sixty–three percent of the samples had a SC less than 15,000; and 90% less than 20,000. The values above 18,000 simply represent instances where, for one reason or another, the person failed to drink water as required. But it is unlikely that any group record would be much better than this.
It is obvious that with a normal daily input of mineral solids equivalent to say 18 grams of NaCl, the daily ingestion of 1.5 grams of electrolytes in the formula water actually increases mineral input by 9%. This increased load should naturally be held to a minimum. By nature, anionic surfactants reach a peak of dispersion. But if the dosage is increased thereafter, the degree of dispersion is lessened. Therefore, one works between two sets of opposing forces: the necessity for adding sufficient reagent to obtain maximum dispersion, but not to a point where it would begin to "salt out" the system. The optimum dosage range of 750 to 1,250 ppm (of a 1:3 electrolyte) agrees with one's expectations, based on overall findings. In particular, the curves shown on
Fig. 25.
It should be noted that even the most effective 1:3 electrolytes we have employed thus far are barely capable of controlling the "really difficult" PVC of long standing. This balance is so delicate that the dosage should be carefully adjusted by frequent electrocardiograms and examinations of the sclera.
It is believed that safe reagents can be found which will have more pronounced electronegative effects on human albumin than the electrolytes we have thus far employed. This is a long and tedious job of reagent screening. Actually, the gastric juice brings all ingested foodstuffs to a pH range of 1 to 3. Therefore, test reagents should also be acidified to say pH 1.5, then elevated to pH 7.4–8.4 with caustic soda. It is quite possible that some reagents would lose their dispersing power at a pH of 1.5, and fail to regain it when the pH is subsequently raised.
BASIC CAUSES OF INTRAVASCULAR COAGULATION
AND CARDIOVASCULAR DISEASE
From the standpoint of Physical Chemistry, we now present our original speculative hypothesis concerning the cause, prevention and relief of intravascular coagulation and cardiovascular disease. Two years ago, this evolved into a working hypothesis. It can be expressed quite simply.
We believe that cardiovascular disease is brought about mainly through inability of the kidneys to adequately remove the abnormally high and ever–increasing mineral solids content of the blood. This increase is due to the ingestion of foodstuffs, which are abnormally high in "salts."
Now broaden the picture. Instead of considering the load simply as "mineral solids content," consider it on the "ionic strength function" basis of Lewis and Randall, wherein valency is squared. Thus, as one progresses from 1:1 to 2:1 to 3:1 type electrolytes, ionic strength increases in ratios of 1:3:6.
Now expand this concept further to the basic work of Schulze and Hardy, von Helmholtz, et al. From mono–, to di–, to trivalent cations, our ratio progresses as 1:15:600. Thus if we have a certain coagulating power from 3,000 ppm of sodium, we can double it by as small a quantity as 200 ppm of calcium or 5 ppm of aluminum. These enormous changes in stability can occur without noticeable changes in Specific Conductance. And so, at 12,000 micromhos, one can obtain no greater accuracy than ±100 micromhos. A change of this magnitude for a 1:1 electrolyte would be insignificant. But such a change in SC due to CaCl2 would be considerable; and due to AlCl3 overwhehning.
The writer does not wish to belabor the Point, but he feels strongly that "trivalent cations," and "salting out" play by far the most important role in the overall problem of blood stability and cardiovascular disease. Up to the present, these aspects have been totally ignored by those who control our food and beverage processing.
But to return to the kidney. With the limited amount of water available to the kidneys each day (under our present habits of water intake), this requires a concentration gradient from blood to urine (as we have previously noted) of 1:2 or 1:3, i.e. 12,000 to 24,000 or to 36,000 micromhos. Apparently, this represents an overload for which the kidneys were not designed. it is difficult to establish even approximately when this overload began — or its extent.
Probably, the extent cannot now be established without making a comparison of the urine and blood electrolytes of "primitive man," with the electrolytes of a typical group of Americans today. Within the past two or three decades, "primitive man" is becoming much harder to find.*
[ * Due to greatly improved transportation facilities within the last few decades, even "semi–primitive man" is becoming much harder to find. Sr. Juan Compte of the Barcelona Water Department advises us that the Andalusian Gypsies of Southern Spain, who still live in rock–hewn caves, now have a refrigerator on the main ledge and a TV antenna protruding through the roof. One can imagine they have, in addition, such items as American processed table salt, and some American processed baking powder containing sodium aluminum sulphate. ]
We can only surmise that this increased use of mineral salts in foodstuffs began in this country about the turn of the century. (Actually, it could have been decades to centuries before.) For many years, the writer has felt that a simple comparison between the SC of freshly–spun blood serum from patients on a coronary ward, and a group of non–coronary patients, would be enlightening. We have no real standards for the SC of blood; and even Albritton's values may not be too representative. A most informative experiment would be to place a small group of persons on a mineral salt intake of, say, 5 grams total (equivalent NaCl), and plot the lowering and leveling off of SC in the blood and urine. We may never ascertain when our overload of "salts" began, but it is an inescapable fact that this overload is the direct result of failure on the part of those concerned with nutrition and food processing to recognize that excessive
electrolytes will "salt out" substantially any colloid system** — including the blood system.
[ ** With proper technique and a suitable microscope, this statement can be verified with dozens of systems. ]
According to Food Technology,* and to Chemical and Engineering News,* the FDA regulations permit a food processor to add substantially any amount of sodium chloride.
[ * For a list of chemical reagents permitted by the FDA, see an article in Food Technology—Sept. 1964, Vol. is, No. 9—entitled: "Chemicals: Twenty–five Years of Progress" (pp. 131-135). See, also, Chemical and Engineering News Vol. 44, No. 42, October 10, 1966—Part I of an article entitled "Food Additives" (pp. 100-120). Also, see Part II in the succeeding issue—Vol. 45, No. 43, Oct. 17, 1966. ]
Food processors add excessive "salts" (excessive over that normally provided by Nature) to improve so–called "taste" of the product. This is an effort to produce something from a can or package that is tangy, palatable and attractive. Food processors cannot be blamed for chemical additions that increase sales. The blame lies with the authorities who permit violation of the simple law of "salting out" — known to every industrial chemist. Moreover, our health authorities should be more cognizant of the effect of trivalent cations upon the stability of any colloid system.
Insoluble aluminum compounds such as Al(OH)3 will dissolve in the stomach through reaction with the HCl of the gastric juice — and the writer rejects any concept that they cannot diffuse directly into the bloodstream.*
[ * The fact they have not heretofore been found to a "significant extent" in the human system does not mean that they cannot be found, if one searches in the right places. ]
These aluminum compounds are permitted by the FDA with little or no restriction, in pharmaceuticals and foodstuffs. Moreover, aluminum cooking utensils are employed extensively in the home, and in commercial food preparation, as well as in beverage preparation and packaging.**
[ ** Examples of packaging are beer and soft drinks in aluminum cans; and "T. V." dinners and snacks packaged in aluminum plates — wrapped in aluminum foil. ]
Those examined by the writer, upon standing in contact with distilled water at room temperature, often will increase the water's Specific Conductance to 6–15 micromhos in a few hours, or 30–50 micromhos in a few days. And some will produce sizeable precipitates.
Our Fig. 167 (p. 245) shows that ovalbumin provides a tremendous bulwark against coagulation by strong cationic electrolytes such as AlCl3. It seems likely that human albumin performs the same function, and in similar manner. But apparently at age 45 to 65, this bulwark is sufficiently overridden* to permit serious intravascular coagulation.
[ * The detail mechanism through which "resistance to coagulation" is lost, should be investigated thoroughly. Once this resistance is lost, it apparently cannot be regained. ]
Doubtless this progression is: ischemia, cardiac infarction — and, in the extreme, necrosis. One cannot fail to be impressed by the findings of Selye and Bajusz on this subject.
We think reduction or elimination of both arrhythmia and the PVC by the reagents and distilled water employed in our Regimen, is due to lowering intravascular coagulation. This, in turn, permits more effective transport and transfer of oxygen, and removal of metabolic products. We also think the PVC and several other types of atypical beats, in addition to being prematurely timed, are associated with failure of the heart valves to close properly and/or seat. We assume this failure to close properly is due to ischemia, which eventually imparts to them a sort, of fibrous and inflexible quality.
With regard to the rhythm of the heart, we quote from Burton's Physiology and Biophysics of the Circulation (Ref. 11-28):
The beating of the heart is intrinsic, independent of all nervous control, which merely modifies the rhythm; it does not initiate it. If kept supplied with oxygen and nutrients, a completely isolated mammalian heart will continue to beat just as does the isolated frog or turtle heart.
All of the nervous and muscular tissues of the heart are capable of independent rhythmic activity, not merely the cells of the cardiac pacemaker (located in the wall of the right atrium), which normally dominate cardiac activity. If conduction from the pacemaker area is cut off, as in the classic experiment of tying a Stannius ligature between the atrium and the ventricles of the frog heart, the heartbeat does not cease, but a new, slower rhythm results from a new "pacemaker" (in the region of the sinoatrial node), which takes up the role of the original pacemaker.
Electron microscopic studies now show that the heart tissue is, rather, a collection of individual muscle or nerve cells in intimate contact with one another, each cell having its cytoplasm separated from that of its neighbors by true cellular membranes. By incubation of minced–up heart tissue of young rats with the enzyme trypsin, which breaks down the substance binding the cells together but leaves the individual cells intact, suspensions of separate cardiac cells can be prepared and cultured in nutrient material. In dilute suspension, the cells are spherical and not in contact; but in a few days of culture on a glass plate, they assume a lenticular shape with elongated filamentous processes. Some of these isolated cells (a few per cent) begin "beating" with rhythmic contractions but with independent rhythms. As the cells multiply and grow, they establish contact with one another. At this time, it is seen that areas of many cells in contact beat in unison, evidently paced by an individual cell. Finally, all the cells of a culture beat together.
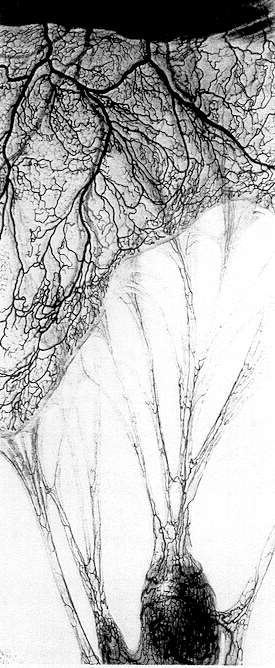
Figure 184, from the Bayne–Jones paper of 1917 ("Blood Vessels of the Heart Valves" — American journal of Anatomy, 21, 449) shows the tremendous vascular structure of a human mitral valve. It would seem that blood in a fluid (rather than agglomerated) state would be able to impart tone and flexibility to this valve, thus aiding in its proper closing and seating. It should be noted that there has been much disagreement over a long period as to whether or not the valves of the heart contain a vascular system.
Figure 184 pictures the elegant drawing by Max Brödel. Johns Hopkins Medical School kindly loaned us the original for this reproduction. It is a composite made from photographs of dye–perfused mitral valves from 14 autopsies.
It is more difficult to appraise the elimination of arrhythmia unless one simply attributes it to improved oxygen transport and transfer — to the nerves per se.
Although death from cardiovascular disease is more prevalent between the ages of 45 and 65, there is every reason to believe that this is just the peak of a gradually worsening physicochemical sequence — one that is observable from the age of 22. According to an article in the New York Times Magazine (January 16, 1966, p. 16), C. P. Gilmore states:
"Autopsies performed on 300 soldiers in Korea — average age 22 — showed that 75% of them had definite early arteriosclerosis."
Mr. Gilmore has assured me his information was obtained from the most authoritative sources.
In 1957, Dr. Robert Kehoe (Director of the Kettering Laboratory, Department of Preventive Medicine and Industrial Health, College of Medicine, University of Cincinnati) and his associates gave aluminum a clean bill of health. This was a literature survey, financed by the Aluminum Association, and published in the AMA Archives of Industrial Health (Vol. 15, No. 5, May 1957) under the title of "Aluminum in the Environment of Man." They note:
p. 367) That aluminum hydroxide dissolves readily in cold HCl at pH values below 4.
p. 369) That the physicochemical characteristics of aluminum compounds can display themselves in (test) animals, but that much greater quantities (of aluminum) are required for such manifestation than are likely to be ingested.
p. 375) That the consumption of canned foods increased in the U.S.A. from one billion 500 million cans (20 cans per person) in 1900, to 23 billion cans in 1955 (140 cans per person).
P. 377) That aluminum is not corroded by canned vegetables unless the media is acid or alkaline.
p. 381) That according to the U.S. Food and Drug Administration, aluminum foils and other aluminum containers are ideal (!)
p. 381) That sodium aluminum sulphate is employed in baking powder.
p. 382-3) That aluminum salts are used in the treatment of both beer and water*
[ * The writer once called to the Public Health Service's attention, a waterworks which was passing (to consumers) about 80% of the applied alum dosage. While this is quite unusual, it did happen. The PHS did not even investigate the matter. ]
p. 399) That the lethal oral dosage of aluminum acetate for test animals is 5 to 15 g/kg; and aluminum chloride, 1 to 3 g/kg.
P. 404) That acute and even fatal poisoning of test animals can result from subcutaneous injection of aluminum sulphate (100 mg/kg).
p. 405) That dissolved aluminum compounds cannot be injected in the bloodstream except in very dilute forms (1% or less) — else precipitation occurs in the bloodstream.
p. 412) That aluminum hydroxide gel, 4 to 6% Al(OH)3, is administered as an antacid with apparent success at a dosage of 4 to 8 ml taken every 2 to 4 hours. (They state that this is equivalent to a maximum possible administration of 2.07 grams aluminum/day.)
The report closes (p. 415) with a statement to the effect that the public and the producer need have no concern about the hazards to public health from widespread usage of aluminum products. They not only felt sanguine about the situation now, but saw no change in the future.**
[ ** The writer does not question the sincerity of this report, but he seriously questions its factuality and believes that it should be reappraised. It should be recalled that prior to the findings of Walter Reed and others, the role of the mosquito was not properly recognized. ]
We believe the Kettering Laboratory report should be critically reviewed in the light of Zeta Potential, and we intend to pose certain questions.
The report employs the terms "aluminum sulphate" and "aluminum chloride" without stating whether they refer to the anhydrous or the crystalline material. We will assume the crystalline; that is, aluminum sulphate with 18 waters of crystallization (MW 660), and aluminum chloride with 6 (MW 241). The conversion factor for the sulphate to the chloride (for equivalent aluminum) is therefore 241 X (2 ÷ 660) = 0.73. Thus, 100 mg/kg of aluminum sulphate is equivalent to 73 mg/kg of aluminum chloride. The report states that:
a.) The lethal subcutaneous dosage of aluminum chloride for test animals approximates 73 mg/kg.
b.) The lethal oral dosage of aluminum chloride (for test animals) approximates 1,000 to 3,000 mg/kg.
c.) Aluminum hydroxide is soluble in HCl at pH values less than 4.
d.) The oral dosage of aluminum hydroxide, that can be taken as an antacid with apparent safety is as high as 2.07 grams per day of aluminum, which is equivalent to 18.5 grams/day of aluminum chloride.
We consider germane to our position in this matter, the following observations and questions:
1. As a broad generalization, the FDA re quires a safety factor*
[ * FDA regulations, under the heading "Safety Factors To Be Considered," state: "Except where evidence is submitted which justifies use of a different safety factor, a safety factor in applying animal experimentation data to man of 100 to 1 will be used. That is, a food additive for use by man will not be granted a tolerance that will exceed 1/100th of the maximum amount demonstrated to be without harm to experimental animals." It is true of course, that certain extensively used items such as sodium chloride are employed at concentrations much higher than would be tolerated by this 1:100 ratio. Again having in mind Schulze and Hardy, the writer finds it difficult to apply these lower standards to either 2:1 or 3.1 electrolytes. Furthermore, it is probable that in the case of aluminum hydroxide antacid, the real factor of safety depends upon the quantity of gastric juice (and its HCl) that is in the stomach at the time — for converting the Al(OH)3 to AlCl3 — But it would also seem that an antacid would not "live up to its name" unless it did exactly that. The writer questions whether any aluminum compounds should be employed in foodstuffs or medications. ]
of about 100. That is to say, a dosage of applied drugs and/or chemicals could not exceed about 1/100 of that amount which (under comparable circumstances) would produce a detectably harmful effect. This criterion would therefore limit the oral administration to 1 ÷ 100 X 1,000 = 10, to 1 ÷ 100 X 3,000 = 30 mg/kg of aluminum chloride. If we assume a human of 180 pounds = 82 kg, this limit would range from 10 x 82 = 820 mg, to 30 X 82 = 2,460 mg. Yet the dosage of aluminum hydroxide (as equivalent aluminum chloride) apparently considered by the Kettering report to be safe is 18,500 mg, which is 18,500 ÷ 820 to 18,500 ÷ 2,460 = 22.5 to 7.5 times that considered safe by the FDA. Instead of a safety factor of 1:100, it must therefore be as low as 1:5 or to 1:14.**
[ ** 100 ÷ 22.5 = 4.5, say 5. 100 ÷ 7.5 = 13.5, say 14. ]
The question: Is this safe practice?
2. Now viewing the matter from another aspect: If the Kettering subcutaneous lethal dosage for animals of 73 mg/kg is applied to the 82 kg human this would approximate 82 x 73 = 6.0 grams of aluminum chloride. Thus, with a possible oral administration of 18.5 grams, death of the human would result if 6.0 ÷ 18.5 = 32% absorbed from the stomach to the bloodstream. The question: Is this any suitable safety factor? If one computes the amount of beverage alcohol that can be found in the blood after taking one drink (or several), it will be found that (according to Albritton's statistics) something like 5 to 13% does pass.
If one uses the Kettering figures of 73 mg/kg lethal subcutaneous, and 1,000 to 3,000 mg/kg lethal oral, one finds that 73 X 100 ÷ 1,000, to 73 X 100 ÷ 3,000 = 7.3% to 2.4% of aluminum chloride does pass. The question: With 2 to 7% of aluminum chloride known to pass from the stomach to the bloodstream, and a passage of 32% known to be lethal, do we have any adequate margin of safety? Do Kettering and FDA consider this safe?
3. One more approach: In the past, many serious scientists must have wondered how the human body can ingest up to 18.5 grams of (equivalent) aluminum chloride and not be killed immediately. We believe that our Zeta Potential curves do much to answer two basic questions: First, why does only 2.4 to 7.3% of ingested aluminum chloride pass from the stomach to the bloodstream? And second, what is the real resistance of the blood to coagulation by a 3:1 electrolyte, In Chapters 8 and 10 (pp. 25 and 32), and on
Figs. 25 and 26 (pp. 34 and 37), we showed that the applied reagent must be considered in two parts: that which remains in the bulk and forces the adsorption; and that which is then adsorbed on the colloid. The passage of aluminum chloride from the stomach to the bloodstream must represent that which remains in the bulk — hence is free and available for this absorption. The major portion (92.7 to 97.6%) must adsorb on the colloids in the stomach and in the small intestine, and in particular on the villi protruding from the walls of the small intestine, which are known to have a tremendous surface area. But we again note the importance of the condition that the daily absorption of aluminum from the stomach into the bloodstream is undoubtedly limited to the quantity of aluminum chloride that can be produced in the stomach over a period of 24 hours. This production is due, of course, to the reaction of aluminum hydroxide with the (natural) hydrochloric acid of the gastric juice.
Our Figure 167 (p. 245) indicates that albumin (or an albumin–like protein) is the real bulwark against coagulation of the blood. In the presence of 3.2% albumin, the dosage of aluminum chloride required for this coagulation was between 1,000 and 2,000 ppm (say 1,500 or 1.5 grams/liter). If we assume that our aforementioned 82 kg individual would have 6 liters of blood, then his albumin would require 6 X 1.5 = 9 grams of aluminum chloride to coagulate. This value checks very closely with the 6.0 grams (82 x 72 ÷ 1,000) set forth by Kettering. We are not attempting to make aluminum the "whipping boy" for heart disease, but we believe that its apparent position in the overall picture of blood coagulation should be made clear. And all the suspected causative factors must be explored if the answer is to be found why 975,000 persons die yearly of cardiovascular disease.
It is unlikely that within the foreseeable future, one will be able to state definitely, why cardiovascular disease becomes lethal principally in one's 50's and 60's. Why don't more succumb in their 20's and 30's? Surely, the basic causes are there.
The answers would seem to lie along one or more of the following speculative hypotheses. By the time, we have reached the age of 50 to 60:
1.) There is a significant reduction in the "buffering" properties of the albumin. (That is to say, the albumin becomes less able to resist the action of excessive 3:1, 2:1 and 1:1 electrolytes.)
2.) There is a marked reduction in the quantity of albumin.
3.) There is a gradual breakdown and deterioration of the organs which produce the albumin.
4.) Platelets have become "leaky" and discharge small amounts of their cationic polyelectrolyte into the system.
5.) The mechanism for "de–fusing" the platelets begins to deteriorate, which also results in leakage of small amounts of cationic polyelectrolyte into the system.
When one tries to go further than this, one encounters the still greater enigma of why all plant and animal life "grows old."
We repeat that the Kettering Laboratory and the FDA should reexamine their concepts in the light of Zeta Potential. In October 1966, we brought these facets to the attention of the head of the New York office of the Aluminum Association, who spent the day with us at our laboratory in discussing these matters. We also took up this matter with certain other officials in the aluminum field.
We again refer to the two–part article in the October 1966 issues of Chemical and Engineering News (a publication of the American Chemical Society.) We excerpt briefly from Part 1, as follows:
1.) The dollar value of food additives increased from 172 million dollars in 1955, to 285 million dollars in 1965, a gain of 66%. Food additives rose from 419 million pounds in 1955, to 661 million pounds in 1965. This is equivalent to 3.4 pounds / person / year, or 4.2 grams / person / day. (This does not include such items as sodium chloride.)
2.) The food industry is the largest single industry in the country. It is the maximum item of the family budget. Retail sales amounted to approximately 80 billion dollars in 1966.
3.) The public is becoming increasingly interested in more sophisticated, flavorful and exotic foods — less bland and more "highly formulated."
4.) Consumption of quick–frozen foods rose from 0.3 billion pounds in 1954, to 2.0 billion pounds in 1964.
5.) The laws of the Food and Drug Administration do not apply to certain additives that, because of years of widespread use in foods, are "Generally Recognized As Safe" by experts in the field. This category, abbreviated as GRAS, includes: salt, pepper, cinnamon, baking powder, citric acid, monosodium glutamate, mono– and diglycerides, and about 575 other materials. (The September 1964 issue of Food Technology [pp. 131-134] lists under the classification of GRAS: vinegar, salt, sugar, aluminum ammonium sulphate, aluminum sodium sulphate, aluminum potassium sulphate, calcium citrate, calcium phosphate, citric acid, potassium bicarbonate,
potassium citrate, sodium acetate, sodium aluminum phosphate, sodium citrate, aluminum sulphate, papain, sodium phosphate, sodium tripolyphosphate. The list under the classification of "Additives" with stated tolerance and/or "restrictions" includes: sodium hexametaphosphate; sodium metaphosphate; sodium phosphate; mono–, di–, and trisodium pyrophosphate; sodium tetrapyrophosphate.)
6.) Chemical and Engineering News notes that "on the basis of somewhat tortured reasoning," these GRAS substances are not even considered food additives under the terms of the Food Additives Amendment.
7.) As of June 1966, about 2,430 food additives (not including GRAS additives) were subject to FDA regulations.
8.) Says Dr. Henry F. Smyth, Jr., An Administrative Fellow at Mellon Institute: "There is no such thing as a class of substances that are poisons. . . . A poison is simply too much."
In closing this section on foodstuffs, we present
Table No. 31 which shows
pH and
Specific Conductance for seventeen canned vegetables and fruits (selected at random), compared with fresh vegetables and fruits. These foodstuffs were first put through a "juicer"* to bring them to a pulp;
[ * Panasonic model MJ-15. ]
then centrifuged at 30,OOO G. to separate the liquid from the pulp. For the vegetables, to which apparently some type of salt had been added to improve taste and/or sales, the increase in Specific Conductance ranged from a factor of 1.45 to 3.75, with an average of 2.54. From
Table No. 26, we concluded that the kidney was being overloaded by a concentration factor of 2 to 3. It would seem that mineral salts in our processed (canned) foodstuffs are being increased by about this same amount. It is significant that when we divide the SC of today's much more concentrated urine, by the factor of 2.54, we obtain the values shown in Table No. 30.
|
TABLE NO. 30
|
|---|
Present SC of Urine
in micromhos
|
Present SC of Urine
in micromhos —
divided by 2.54
|
|---|
|
20,000
|
7,900
|
|---|
|
2,5,000
|
9,900
|
|---|
|
30,000
|
11,800
|
|---|
|
35,000
|
13,700
|
|---|
Therefore, it would seem that if a person ingested only fresh vegetables without salt (with any reasonable amount of water), his kidneys would operate in the 5,000 to 15,000 micromhos range — not 25,000 to 35,000.
It is of interest that the Specific Conductance of fruits is considerably lower than vegetables, ranging from 2,000 to 7,100 for the series listed. Apparently, this is simply the nature of these foodstuffs. In no instance was there a rise in SC upon processing (canning). Conversely, two values shown represent a decrease. This is because a characteristic of fruits is sweetness; therefore the processors add sugar instead of salt. Sugar is of course nonionic, and it impedes the travel of dissociated ions — thereby reducing the apparent Specific Conductance.
These figures may not impress the reader, but we doubt that many anthropologists would be surprised at the failure of the kidney and heart under these circumstances.
In closing this chapter on the basic causes of intravascular coagulation and cardiovascular disease, it should be noted that "salting out" and the effect of highly coagulating electrolytes are not the only factors contributing to cardiovascular disease. The medical profession has repeatedly pointed to table salt, alcohol and tobacco — in excess — as traceable causes. Selye has also pointed strongly to "stress." We are in full accord with these points of view. We are also in complete agreement with Dr. Paul Dudley White's statements on the value of proper exercise and nutrition in maintaining the cardiovascular system in good condition.
|
TABLE NO. 31
|
|---|
|
SPECIFIC CONDUCTANCE AND pH OF CANNED v.s. FRESH VEGETABLES AND FRUITS *
|
|---|
|
Vegetables and Fruits
|
pH Fresh
|
pH Canned
|
SC Micromhos Fresh
|
SC Micromhos Canned
|
Concentration Factor
|
Percent
Increase
|
|---|
|
(1)
|
(2)
|
(3)
|
(4)
|
(5)
|
(6)
|
(7)
|
|---|
|
1.) Asparagus
|
6.1
|
5.5
|
4,800
|
18,000
|
3.75
|
275
|
|---|
|
2.) Beets
|
5.9
|
5.6
|
5,200
|
17,500
|
3.36
|
236
|
|---|
|
3.) Peas
|
6.8
|
6.2
|
11,000
|
17,500
|
1.60
|
60
|
|---|
|
4.) Lima Beans
|
6.3
|
5.3
|
7,400
|
21,000
|
2.84
|
184
|
|---|
|
5.) Corn
|
6.3
|
6.2
|
4,700
|
12,000
|
2.56
|
156
|
|---|
|
6.) Tomatoes
|
4.3
|
4.2
|
6,800
|
13,500
|
2.00
|
100
|
|---|
|
7) Carrots
|
6.3
|
4.9
|
13,000
|
19,000
|
1.45
|
45
|
|---|
|
8.) String Beans
|
6.3
|
5.3
|
7,400
|
21,000
|
2.84
|
184
|
|---|
|
Average
|
—
|
—
|
7,540
|
17,400
|
2.55
|
155
|
|---|
|
9.) Black Grapes
|
—
|
—
|
3,400
|
—
|
—
|
—
|
|---|
|
10.) Red Grapes
|
—
|
—
|
3,600
|
—
|
—
|
—
|
|---|
|
11.) Bananas
|
—
|
—
|
7,100
|
—
|
—
|
—
|
|---|
|
12.) Grapefruit
|
3.6
|
3.2
|
2,900
|
—
|
—
|
—
|
|---|
|
13.) Pears
|
4.0
|
4.2
|
2,300
|
1,100
|
—
|
decr.
|
|---|
|
14.) Oranges
|
4.0
|
—
|
4,000
|
—
|
—
|
—
|
|---|
|
15.) Apples
|
4.4
|
3.4
|
2,000
|
1,600
|
—
|
decr.
|
|---|
|
16.) Strawberries
|
3.6
|
—
|
4,000
|
—
|
—
|
—
|
|---|
|
17.) Watermelon
|
—
|
—
|
2,700
|
—
|
—
|
—
|
|---|
|
Average
|
—
|
—
|
3,600
|
—
|
—
|
— |
|---|
[ * Note that the SC of fresh vegetables averaged about 7,500 micromhos, and fresh fruit about 3,600. It is obvious that a person substituting fruits for vegetables would halve his mineral solids input. In this connection, it would also seem probable that produce from "organic" gardening would be lower in mineral solids than produce raised with conventional fertilizers. ]
It should be stressed that in many instances, a large increase in the SC of a processed food (particularly a canned food, as above shown) is due principally to the addition of sodium chloride. Both Bajusz and Selye (Refs. 11-39, 40 & 41; 11-13 & 14) outline the role potassium plays in cardiovascular systems and disorders. They emphasize that heavy additions of sodium to the diet deplete potassium (by increasing its output in the urine), and often result in high blood pressure, myocardial infarction, necrosis, and even death. When one critically examines the U. S. Dept. of Agriculture Handbook #8 (Ref. 12-41), one finds that potassium — not sodium — is the principal cation in most raw (natural) foodstuffs. Our arbitrary use in food processing of sodium (rather than potassium), and in quantities far exceeding the original mineral content of the foodstuff, is improvident and presumptuous. It reflects a flagrant disregard of natural laws — which cannot, with impunity, be violated. Moreover, we believe that all "salt" added to foodstuffs in processing — and "salt" sold by the grocer for table and cooking use — should consist of a mixture of 60% potassium chloride (or sulphate), and 40% sodium chloride. This would tend to maintain the original K / Na balance of the foodstuff, and also reduce the addition of sodium by about half. Unless remedial and control steps of this order are taken in food processing and preparation, indications are that the death rate from cardiovascular disease will rise. Contributing to this trend is the increasing usage of chemical additives, and also the increasing usage of aluminum for food packaging.
PREVENTION AND CONTROL OF INTRAVASCULAR COAGULATION
AND CARDIOVASCULAR DISEASE
PREVENTION
Obviously, in cardiovascular disease (CVD) the aim should be prevention through elimination of the basic causes. Control measures should be considered simply a temporary expedient, principally for those now over 40. Even allowing for persons who drink or smoke excessively, the writer believes the death rate from CVD could be halved in one year, and reduced more than 90% in a decade. But this of course would require positive action on a national scale.
Unfortunately at the present time, the individual does not become fully aware of CVD until he has experienced some sort of attack — such as severe angina or paroxysmal tachycardia — and of course at this stage, the proper time to institute preventive measures has long since passed.
In the writer's opinion, realistic, large–scale prevention can be readily effected — but only through appropriate Food & Drug Administration regulations.
Only a few would be necessary and I list them as a "public service." (But I suspect there is little likelihood they will be employed.)
1.) As an indication of the degree to which the "salt" content of a foodstuff has been increased through processing, all containers should be marked with a green, yellow or red stripe. For a green stripe, the SC of the processed food should not exceed the original value by more than 10%. For the yellow stripe, 25%. Increases over 25% should be marked with a red stripe.
2.) All beverages, including milk, should be similarly coded — with the green, yellow and red criteria representing actual SC values of: zero to 750; 750 to 1,500; and over 1,500 micromhos, respectively.
3.) The label on all foodstuffs and beverages should show (in micromhos) the initial SC, and the final processed SC.
4.) The release of metallic ions from "cans," and from vessels employed in food and beverage packaging, should be controlled by more effective linings and coatings. When empty food containers are thoroughly cleaned and filled with distilled water, and kept under agitation at room temperature, the allowable rise in the SC of this distilled water should be limited to not more than 2 micromhos per day, 10 micromhos in 5 days, or 20 micromhos in 30 days.*
[ * Figure 185 shows the rise in Specific Conductance of distilled water added to the ten cans containing the eight vegetables and two fruits shown on
Table No. 31. Also shown is an aluminum beverage can. We assume that conductivity is caused by metallic ions, but of course, their nature should be determined by a spectographic analysis. ]
The concentration of the cation in the distilled water after standing 5 days at room temperature should not exceed 3 ppm when analyzed by spectrographic or other approved methods.
5.) No utensils for the preparation, handling or storage of food that permit trivalent cations to be discharged to the foodstuff should be manufactured, employed or offered for sale. The standards set forth in item 4 (above) should apply.
6.) No trivalent or quadrivalent cationic electrolytes (or cationic polyelectrolytes) added to food or beverages should show a residual in the finished product to exceed 0.10 ppm, expressed as the cation.
7.) The addition to beverages, foodstuffs and medications of electrolytes containing trivalent or quadrivalent cations (employed for purposes other than coagulation) should be prohibited. Aluminum sulphate should be permitted for coagulation — but not in quantities to exceed 100 mg/l or 200 mg/kg of system. Here, care should be exercised to insure maximum precipitation of the aluminum as a hydrate, followed by removal of this sludge from the product.
In the light of our overall findings, we believe that all existing food additives should be re–screened with respect to Zeta Potential. All additives with dangerous cationic properties should be prohibited — now and in the future.
The public should be adequately appraised of the effects on the human system of excessive 1:1, 2:1 and 3:1 electrolytes. Those who still want food "tangy" with salt would be free to purchase them, and manufacturers would then plan their production accordingly.
CONTROL
In the main, intravascular coagulation is related to an insufficiency of water intake. Therefore, the writer cannot envision control without use of water. As we have stated before, if any of the dispersing agents we have discussed were taken in capsule form without water, intravascular coagulation would be actually increased, not decreased.
It is almost superfluous at this point to go into greater detail on how dispersing agents break up and/or prevent intravascular coagulation. It is obvious that they render the system more disperse through increasing mutual repulsion, and that albumin (or an albumin–like protein) serves as a veritable bulwark against sudden or accidental change that would threaten the integrity of the organism. We therefore present without comment the basic dispersion curves for heparin, potassium citrate,*
[ * We do not believe that the potassium citrate employed in our Regimen has any significant relationship to the KREBS (citric acid) cycle (see pp. 59-61, Ref. 12-40). The potassium (or sodium) citrate in our Regimen functions because of strong electronegative Zeta Potential, and this is due to the trivalence of its anions. Other trivalent anions (or anions of equivalent valence) are almost equally effective. ]
sodium hexametaphosphate, and low molecular weight dextran. ( Figs.
187,
188,
189,
190, ) Dextran is now employed in medicine as a dispersant. The curve for sulphates is given on
Fig. 19. It is readily seen that these reagents could be employed for industrial dispersion. Conversely, it is evident that with care in selection, many industrial dispersants could be employed for preventing or correcting intravascular coagulation.
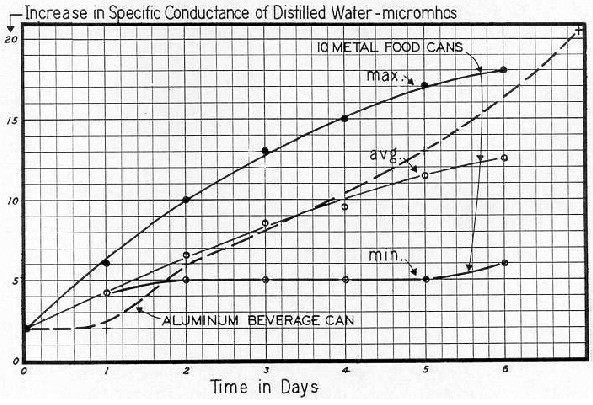
Our Regimen for control of intravascular coagulation poses certain problems that are not easily solved. For example, only distilled water (with appropriate quantities of a dispersing agent) should be employed. Tap water will not suffice. And also, the quantity of water needed is a matter of concern. The writer does not believe it is economically practical for municipal waterworks to produce a water containing suitable dispersing characteristics and furnish it under pressure to the distribution system. This would involve complete deionization, then treating with appropriate reagents the entire required output of 125 gallons per person per day. Since only 1½ quarts of drinking water is needed per person per day, it would seem simpler to have all waterworks throughout the country dispense deionized water in plastic containers at a few cents per gallon on a cash and carry basis. Or, the individual could install his own distillation equipment, as shown.*
[ * With present–day heavy stream pollution, finished waters from municipal sources leave much to be desired from the standpoint of taste, odor, etc. Even with no added electrolytes, distilled water may prove a welcome change. ]
(As previously noted, deionizers are preferred for the removal of mineral salts from large volumes of water. They are not recommended for home use because of the possibility of undetected bacterial contamination.) The individual would thus be free to prepare a water containing a proper dispersing agent for drinking use.
There should be mobile cardiological laboratories in every city in the country where one could, without appointment, have a quick examination and grading of his sclera. We have never observed malfunctional beats at Grade 2 IVC. At Grade 3, malfunctions are sometimes observed, but in the order of only 1 to 5%. At Grade 4 or 5, malfunctions in the range of 10 to 30% — or even higher — are not uncommon. Therefore, control should be instituted at Grade 2. In these mobile laboratories, one should also be able to have his ECG monitored for malfunctional beats.
These are realistic steps that could be taken. In the writer's view there is, unfortunately, no probability of any meaningful program to control cardiovascular disease on a national level in the foreseeable future. Thus, those persons involved who are genuinely concerned, must face the problem individually.
The writer is a research chemist and does not prescribe. This is left to one's physician. But I feel no reluctance in setting forth and sharing with any interested party or group my personal experience and the considerable information that has become available through our research.
We have no way of knowing at this stage how our work will be received by the medical profession — individually or as a whole. During the past year, several physicians became personally acquainted with our research and they seem convinced that we are on the "right track." They are enthusiastic about our results. But, of course, "one swallow maketh not a spring."
The writer felt an obligation to prepare this chapter on blood stability and cardiovascular disease. It was necessary to spend ten years of intensive research to develop the role of Zeta Potential in the stability of industrial colloids. And only after this basic pattern was firmly established, could one hope to apply it successfully to biological systems. Moreover, the application of basic principles of Zeta Potential to the human organism was an ideal test of their verity — because a most important criterion for evaluating any hypothesis involving a natural law is: "Does it obtain (prevail) for biological systems?"
But with Chapter 22, my "obligation to society," as it were, is fulfilled. I would not debate my thesis for a moment with anyone of a different discipline than mine. I am not a reformer, and if a person wishes to ingest 50 grams of "salt" a day, plus 25 grams of a 3:1 electrolyte, I say "less power to him." My satisfaction with what we have accomplished in this field fully justifies the effort expended.
The writer has every confidence that cardiovascular disease will soon be prevented, as well as controlled. If it is not accomplished in this country, it will be in some country where politics and science are less financially intertwined.
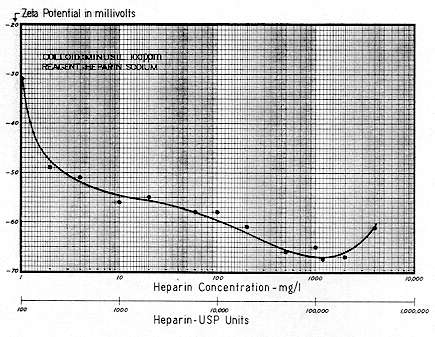
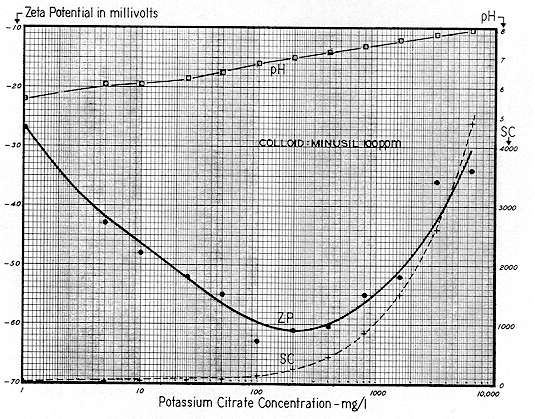
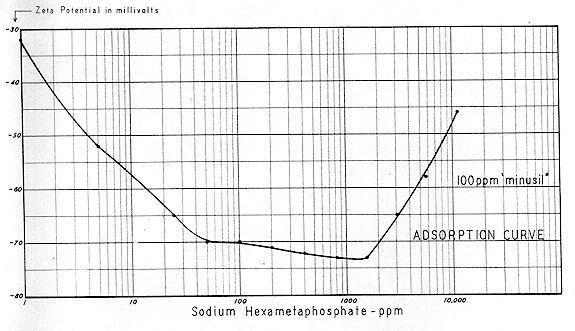
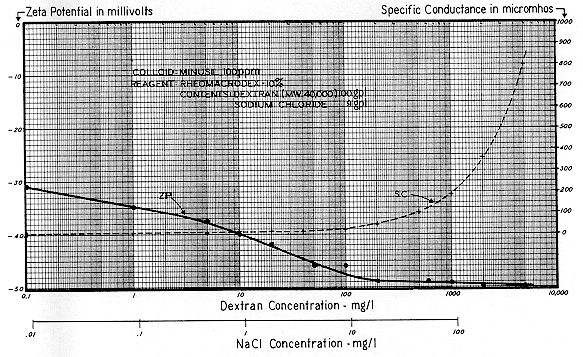
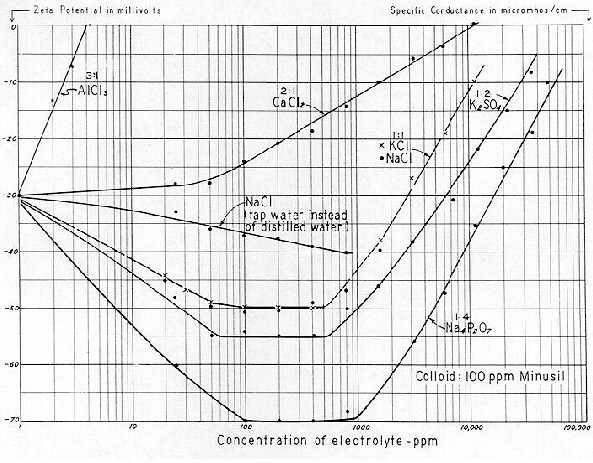
EXAMPLES OF THE REMEDIAL EFFECTS OF THE REGIMEN
EXAMPLE 1 — The Story of Koko and the Aluminum Dish

Left, is a photograph of Koko, the writer's six–year–old Panama Parrot. His I.Q. is high and his talk enlightening after any two–day convention or one–day trip to Washington. He comes to the table each morning for fruit, and again at night for a green salad. His plumage and coloring are excellent, due to well–balanced foodstuffs. For exercise, he is encouraged to fly.
If one is accustomed to "handling" a parrot, particularly a young and healthy bird, one is quite aware that the feet are noticeably warm. At about the time the writer established his Regimen of synthetic water, Koko's feet suddenly grew cold. Then he ceased to fly. Within five days, he lost most of the feathers from an area 5 cm in diameter on his chest, and his down appeared dry and lifeless under the microscope. A competent veterinarian examined Koko. He suggested vitamin deficiency and prescribed B Complex.
 Instead, the writer withheld all vitamins or other forms of treatment, and went to work on Koko's water dish, which was cast aluminum. For test, it was filled with distilled water plus 100 ppm of Minusil. In about 24 hours the ZP of the system progressively changed from –30 to +15 mv. The dish was probably "cast scrap," and in corroding, undoubtedly released aluminum ions at a more rapid rate than normal for kitchenware. The writer concluded that Koko had simply become another victim of intravascular coagulation, and was on his way to a genuine heart attack. He joined our group and was placed on the Regimen, with no change in the formula — which at that time was 1.0 gpl of combined sodium and potassium citrate, half and half.
Instead, the writer withheld all vitamins or other forms of treatment, and went to work on Koko's water dish, which was cast aluminum. For test, it was filled with distilled water plus 100 ppm of Minusil. In about 24 hours the ZP of the system progressively changed from –30 to +15 mv. The dish was probably "cast scrap," and in corroding, undoubtedly released aluminum ions at a more rapid rate than normal for kitchenware. The writer concluded that Koko had simply become another victim of intravascular coagulation, and was on his way to a genuine heart attack. He joined our group and was placed on the Regimen, with no change in the formula — which at that time was 1.0 gpl of combined sodium and potassium citrate, half and half.
His feet grew warm in 48 hours, and he began to fly in three days. He grew back all his lost feathers in five weeks. The veterinarian thought his recovery was amazing. Koko had drunk from the same dish for more than three years. His delayed intravascular coagulation (of a truly serious nature) may be compared to the death of most persons from cardiovascular disease in their 40's, 50's and 60's, and the case is interesting and significant. Koko's aluminum dish was cycled a dozen or so times with distilled water to bring out the full bloom of the aluminum oxides, and it is shown on
Fig. 191. It is available to any genuinely concerned party for further study.
For "treatment," I did not change his mode of life, except to eliminate his intake of aluminum by substituting a Pyrex dish — and provide him with a water of known dispersing characteristics. It would appear that the blood and albumin of the parrot and human have much in common.
The General Manager of the company who made the cage was very cooperative and promised to immediately change all his units to glass or plastic feed dishes. It is a source of the greatest satisfaction to me that our research enabled us to combine, for the first time, the basic contributions of von Helmholtz and Knisely — and put them to such dramatic and worthwhile use. Koko is (and has since remained) in excellent health, and he is still on the same formula I employ.
EXAMPLE 2 — The Writer's Paroxysmal Tachycardia
As previously noted, the writer has a ten–year history of paroxysmal tachycardia. A medical friend suggested that it would be enlightening for me to describe one of my attacks in detail, from the standpoint of Physical Chemistry and Zeta Potential. Actually, from this standpoint, indications are that one should view paroxysmal tachycardia as a blood disorder — not a cardiovascular condition.
I quote briefly from Chapter 14 of Friedberg's Diseases of the Heart,* which describes this condition:
[ * Ref. 11-24-Published by W. B. Saunders Co., Philadelphia, Pa., Third Edition. ]
The etiology of atria paroxysmal tachycardia is obscure. The same nervous, toxic and digestive disturbances, emotional stress, fatigue, exertion and disease of the biliary tract, which have been held responsible for premature beats, have also been indicated as responsible for attacks of atrial tachycardia. ...
The atrial rate varies usually between 140 and 220 per minute, but most commonly between 180 and 200. Occasionally the atrial rate is as high as 300 per minute. The ventricles almost always respond to each atrial beat.
The onset is sudden and without warning. Often a rapid change in position of the head or trunk, an unexpected emotional upset or an unpleasant dream appears to unleash the attack. But usually there is no apparent precipitating cause.
... Sometimes there is definite precordial pain, a smothering sensation in the chest and throat, fullness or a sense of pulsation in the neck and head or epigastric distress. ...
These local symptoms are often overshadowed by psychic and reflex nervous manifestations, especially when the attacks persist for more than a few minutes. Anxiety or even angor animi, weakness or exhaustion, coldness or sweating, and a sense of smothering may occur early. There may be dizziness, faintness or momentary syncope at the onset. ...
There may be polyuria during or at the termination of the paroxysm.
Serious circulatory disturbances may develop if the rate is exceedingly rapid, but especially if there is underlying cardiac disease and if the paroxysm is prolonged. ...
Weakness, pallor, perspiration and coldness of the extremities may be due to diminished cardiac output. The clinical picture of shock with extreme hypotension may, develop. ...
... The attack may occasionally terminate fatally, either with signs of progressive failure or suddenly. As a rule, however, the paroxysm ends abruptly, normal rhythm is restored and the symptoms and signs of circulatory insufficiency abate rapidly. ...
In severe and prolonged attacks the patient may appear obviously anxious or agitated and there may be objective signs of congestive failure.
The pulse is very rapid and small. ...
Paroxysms of atrial tachycardia stop as abruptly as they begin, either spontaneously or because the patient adopts certain positions, takes a deep breath, holds his breath with glottis closed, or because the physician controls the attack. As a rule, the attacks subside spontaneously in seconds or minutes, but attacks with disturbing symptoms last for several hours and occasionally for several days. ...
The physician's most useful procedure is to massage the right carotid sinus and, if this is ineffective, the left carotid sinus for 10 to 30 seconds. ...
A similar vagal reflex may be induced by firm pressure over one eyeball at a time and then if necessary over both eyeballs simultaneously.
Digitalis, administered intravenously, is now widely employed as the drug of choice in the treatment of atrial tachycardia. ...
Ouabain is administered if there is great urgency. ...
Pronestyl may also succeed in terminating attacks of atrial tachycardia although it is generally less effective than in the control of ventricular tachycardia. ...
Potassium salts have been employed to control the atrial tachycardia resulting from digitalis intoxication. ...
Magnesium sulfate, 15 to 25 cc of a 20 per cent solution, injected intravenously, 0.5 cc. at a time, has been effective in the control of paroxysmal tachycardia.
The prevention of frequent attacks of paroxysmal tachycardia is difficult because of ignorance as to the underlying or precipitating cause. All of the measures advised in the treatment of frequent extrasystoles should be employed including the avoidance of emotional tension, fatigue, tobacco, coffee and alcohol, indigestion and constipation, unusual effort or exertion.
It may be that Friedberg speaks from personal experience. Certainly, he gives as good a description of paroxysmal tachycardia as one can obtain. His symptoms are accurate and his appraisal realistic. But he appears undecided when he states: "There may be polyuria (excessive urination) during or at the termination of the paroxysm." I long shared his indecision on what now appears to be a most important point. In fact, in discussing this matter with others who are subject to these unpredictable attacks, I found that they were also undecided. One stated that his extreme fright completely dominated his other sensibilities. I can testify to the reasonableness of that.
This inability of recall put the writer in about the same state as the long–bearded octogenarian who was unable to get a good night's sleep after he was asked: "Do you sleep with your beard under the covers, or on top of the covers?" Therefore, I decided to solve this perplexing question of polyuria, because I believed that the nature of the mineral salts discharged during an attack (compared with normal) might give some clue as to the basic cause of the attack. I therefore kept a graduated cylinder handy, and forewarned an assistant that he might be called upon at any time of the night to operate the oscilloscope. Eventually the attack occurred, and measurements were duly recorded.
Results were rather astonishing.
Figure 193 shows the plot of this data; also that of a subsequent attack about one year later. This represents a current incidence of two attacks in two years, compared with a previous ten–year experience of an attack every month or two.
In collecting this data, I recorded the hour, the time interval between sample collections, and the quantity. From this I computed the rate per hour, and the "equivalent rate" for a 17–hour period. I term this the "rate per day," because 17 hours represents one's normal, wakeful "micturating" day. Values throughout a "normal" day range from about 0.5 to 3.0 liters/day, or an average of say 1.5 liters/day.

Referring to Fig. 193, I was awakened from normal sleep by these attacks, and the slope of the curves indicates that I must have awakened at the actual onset. By finger and thumb pressure on my carotid arteries, it was evident that my pulse rate was about 100 per minute, and that there were numerous malfunctional beats which threw no blood. At the onset of these attacks I put on an overcoat and trousers, and was at my laboratory two blocks away with leads connected and the oscilloscope in service within 20 minutes. I therefore had a continuous cardiographic picture of my electrical output and wave forms.
The ECG and the traces of the radial pulse showed that the average rate of heartbeat 20 minutes after the onset was 190/minute, and pulse rate 105. Therefore, it was evident that 45% of my heartbeats were pumping no blood. On most of the beats where blood was discharged, the pulse traces indicated that the quantity was significantly subnormal.
It is seen from
Fig. 193 that Episode #1 lasted 2 hours and 10 minutes; and Episode #2, exactly 3 hours. In Episode #1, I passed 860 ml of urine in 130 minutes, or 6.6 ml/minute. This was equivalent to an average rate of 6.8 liters/day.*
[ * 6.6 X 60 X 17 ÷ 1000 = 6.8 liters/day. ]
Peak production of 12.5 liters/day was reached about 1 hour and 20 minutes after the start.
In Episode 42, 1 passed 1,600 ml of urine in 180 minutes, or 8.9 ml/minute, an average of 9.1 liters/ day. A peak of 17.5 liters/day was reached in 1 hour and 15 minutes after the start.
During Episode 41, the SC of my composite urine was 9,000 micromhos, equivalent to 4.5 grams of sodium chloride per liter. This (0.860 X 4.5) is equivalent to 3.9 (say 4 grams) of NaCl passed in a period of 2 hours and 10 minutes. During Episode #2, the SC of my composite urine was 8,600 micromhos, equivalent to 4.3 grams of salt/liter. This (1.6 X 3.4) is equivalent to 6.9 (say 7 grams) of salt passed during this 3–hour period.
Assuming my system contains 6 liters of blood at 12,500 micromhos, my blood must contain the equivalent of 6 X 6.25 = 37.4 grams of NaCl.**
[ ** Actually, according to typical analyses shown on
Table 13 blood contains about 9.0 grams of mineral salts per liter, making the factor for conversion from SC to blood minerals 9,000 ÷ 12,500 = 0.72, say 0.70 (as contrasted with 0.50 for NaCl). Conversely, the factor to convert blood minerals to SC should be 12,500 ÷ 9,000 = 1.39, say 1.40 (vs 2.0 for NaCl). However, we will not undertake this refinement in these computations because we are mainly demonstrating principle. We have never determined the factor for urine, but we presume it would approximate that of blood. If so, the grams of equivalent NaCl per liter of blood would be 12,500 X 0.7 = 8.75 g/l — giving an overall mineral content of 6 X 8.75 = 52.50 — say 50 g total This seems reasonable and checks closely with the concentration of 9.08 gpi given in Table No. 13. ]
During Episode #1, therefore, my kidneys removed from my blood 4 ÷ 37 = 11% of my mineral salts in 2 hours 10 minutes, or 1.9 grams per hour. During Episode #2, my kidneys removed 7 ÷ 37 or 19% of my mineral salts in 3 hours. This rate is equivalent 7 ÷ 3 = 2.3 grams/hour.
Thus, for good and sufficient reasons (known best to my glandular system) my kidneys rid my blood of mineral salts at a rate of 1.9 to 2.3 (say 2) grams per hour. Had they kept this up for a 17–hour period, they would have removed 17 x 2 = 34 grams, or substantially the entire mineral content of my blood system.
The question: "Why?"
Naturally, my comments are confined to my own kidneys, about which I can express my own professional opinion. But before discussing the composition of the mineral salts discharged, it should be noted that paroxysmal tachycardia generally has been regarded as an episode of short duration. It would seem that here we have a reason. Even an eight–hour siege at this rate of mineral loss would halve our blood electrolytes — probably producing death through osmotic imbalance.
During Episode #1, I collected a composite urine sample from 1:00 to 3:10 A.M., which I thought would contain the electrolytes my kidneys were endeavoring to "get rid of." I also collected a "catch" sample at 9:00 A.M., 6 hours after the termination of the Episode. My purpose was to determine the norm after the Episode; that is to say, normal after the kidneys had purged these unwanted salts.
Results are shown in
Table No. 32. The constituents of columns 2 and 3 are on the basis of mg/l.
In columns 4 and 5 we show percent of total solids. The latter expression compensates for the difference in the dissolved mineral content of the samples.
One must be cautious in drawing conclusions from such sparse data. But these indications are that my blood was overloaded with mineral salts. It would seem that to produce these reactions, these salts would be of a coagulating nature. And, in addition to 1:1 electrolytes, there also would be an excess of the 2:1 and/or 3:1 types. These salts were probably "tied up" somewhere in my blood system. Judging from the curves of Fig. 167 (p. 245), it is possible that the strong cations could be combined with my albumin. Therefore it seems reasonable that during this "paroxysmal purge" the kidneys would tend to discharge the objectionable cations and anions, but retain the needed ones.
The results of analysis
(Table 32) bear out this hypothesis, because the kidneys:
1.) Discarded the sodium, but retained the potassium
2.) Discarded the calcium, but retained (to a very high degree) the magnesium
3.) Discarded the aluminum, by a 13:1 ratio
4.) Discarded the manganese, lead, silicon and silver
I will not comment on the other elements, as I have no criteria for assessing either their value or their harmfulness. The cations of the heavy metals, whether di– or trivalent, are generally considered undesirable; sometimes dangerous.
It is obvious that the riddance of sodium, but retention of potassium and magnesium, agrees fully with concepts that Selye and Bajusz have stressed for years.
Another point: For the past two years I have attempted to completely avoid intake of
aluminum in any form. But despite this, it is evident that my system continued to ingest it — from sources unknown. Thus, as previously noted, the ratio in my urine during and after this purge was 13:1.
It must be concluded that this intake was due to foodstuffs — either processed with aluminum compounds and/or prepared in aluminum vessels. Both practices are permitted by the FDA. In fact they apparently consider aluminum excellent for packaging.
|
TABLE NO. 32
|
|---|
SPECTROGRAPHIC ANALYSES* OF URINE COLLECTED DURING
(AND 6 HOURS
AFTER) AN ATTACK OF PAROXYSMAL TACHYCARDIA
|
|---|
|
Adjusted Probable Concentration
|
|---|
|
mg per Liter
|
Percent
|
Ratio
|
|---|
|
Element
|
During Attack
|
After Attack
|
During Attack
|
After Attack
|
Ratio Cols. 4:5
|
|---|
|
(1)
|
(2)
|
(3)
|
(4)
|
(5)
|
(6)
|
|---|
|
Sodium
|
1700
|
1800
|
89
|
33
|
3:1
|
|---|
|
Potassium
|
170
|
1800
|
8.9
|
33
|
1:4
|
|---|
|
Calcium
|
17
|
18
|
0.89
|
0.33
|
3:1
|
|---|
|
Magnesium
|
17
|
1800
|
0.89
|
33
|
1:37
|
|---|
|
Aluminum
|
9
|
2
|
0.47
|
0.037
|
13-1
|
|---|
|
Phosphorous
|
9
|
2
|
0.47
|
0.037
|
13:1
|
|---|
|
Iron
|
0.2
|
0.2
|
0.010
|
0.0037
|
3:1
|
|---|
|
Manganese
|
0.2
|
0.02
|
0.010
|
0.00037
|
30:1
|
|---|
|
Rubidium
|
0.2
|
2
|
0.010
|
0.037
|
1:4
|
|---|
|
Lead
|
0.1
|
NF
|
0.005
|
NF
|
—
|
|---|
|
Silicon
|
0.1
|
0.02
|
0.005
|
0.00037
|
13:1
|
|---|
|
Silver
|
0.02
|
NF
|
0.001
|
NF
|
—
|
|---|
|
Copper
|
0.02
|
0.2
|
0.001
|
0.0037
|
1:4
|
|---|
|
Cadmium
|
NF
|
0.2
|
NF
|
0.0037
|
—
|
|---|
[ * Again, the spectrographic analyses were done under the supervision of Dr. Bell of Lucius Pitkin, Inc. ]
Elements checked for but not found:
Zinc, Tin, Germanium, Nickel, Cobalt, Molybdenum, Vanadium, Lithium,
Tungsten, Titanium, Zirconium, Barium, Strontium, Indium, Bismuth, Amtimony, Arsenic, Thallium, Gallium.
[ The Elements in green are required for a healthy body. ]
In rationalizing these episodes of paroxysmal tachycardia, from the standpoint of Physical Chemistry, one may form a speculative hypothesis:
1.) A sensing mechanism (possibly one or more glands) detected that something was wrong — that 1:1 and/or 2:1 and/or 3:1 electrolytes had built up to a dangerous concentration in the blood stream. It therefore activated an alarm.
2.) My cardiovascular system responded to this alarm by increasing the rate of heartbeat, which normally should increase the rate of blood circulation, and transport of oxygen and metabolic end–products.
3.) My sensing mechanism had no way of knowing that the food processors of our "advanced western ecology," through improvement of "taste" (and the FDA, through preoccupation and politics) — had long since "salted out" my blood. Moreover, through ischemia and infarction they had rendered my heart valves fibrous, inflexible and petrifactive. My sensing mechanism was also unaware that my mitral and aortic valves (that once would have seated properly at a high operating rate) would now fail to close properly. They would permit excessive back–slippage at even 100 beats per minute, thus making 150 to 200 beats even more impossible.
4.) But the warning signal went through. My heart action was stepped up, and my valves commenced to malfunction. As my heart beat increased, the valve closure worsened. Thus the increased pumpage which my Sensing mechanism called for, ironically resulted in significantly diminished blood output.
5.) My kidneys also received the danger signal from my sensing mechanism, and they began immediately to discharge to waste the dangerous concentration of mineral salts. However, they apparently sensed that to do this, time was of the essence, and they must operate at their peak efficiency for mineral solids removal. This peak apparently is in the range of 8,600 to 9,000 micromhos, not the 20,000 to 35,000 micromhos at which most kidneys now generally operate because of insufficient water. Thus the rate of liquid output (for a short period of time) reached 12½ to 17½ liters per day*
[ * To obtain this much–needed water, my kidneys could not withdraw it from the blood, for this would have had the effect of concentrating the solids, i.e., decreased the liquid volume by 860 ÷ 6,000 = 15%. Therefore, they must have "borrowed" this water from some other source — say interstitial fluid. We have every proof that the kidneys function more efficiently when discharging a relatively low concentration of electrolytes, but they cannot so operate simply because of insufficient water input. ]
6.) In these two cases of paroxysmal tachycardia, the unwanted electrolytes were discharged in two and three hours respectively, after which the emergency was ended. If our hypothesis is correct, we would judge from the curves of
Fig. 193 that once the paroxysm is triggered, it must run its course. The necessity for additional water, to supply the kidney during this period of very heavy discharge, is clearly evident. It would seem that the ingestion of one or two quarts of distilled water during the paroxysm could be helpful.
7.) We again stress that the 860 ml of water for Episode #1, and 1,600 ml for Episode #2 could not have come from the blood system, because this would have concentrated the blood electrolytes. This water must therefore have been "borrowed" from some other source. (The writer refrained from drinking water during these episodes.) After Episode #2, the writer drank two quarts of water (from 9 A.M. to 5 P.M.) before any significant quantity of urine was discharged. This indicates that the water had been "borrowed," and was being "returned."
In closing this section, it should be noted that through the use of the Regimen, the incidence of my paroxysmal tachycardia has decreased about 80%. And the two episodes discussed herein came after out–of–town trips which interrupted my Regimen for periods of 4 or 5 days.**
[ ** I do not now permit my Regimen to be interrupted. ]
This emphasizes that both heparin and the writer's Regimen should be considered as regulatory and control techniques, not truly curative measures. The writer has no idea how close the paroxysmal tachycardia described conforms to the average pattern — or whether it resembles it at all. But it would seem worthwhile for someone to find out. It can be that paroxysmal tachycardia can be prevented.
UNDER CONSTRUCTION --- More Coming
" Control of Colloid Stability through Zeta Potential "
by Thomas M. Riddick — Introduction
Understanding Colloidal Suspensions
A summary of this book submitted by Frank Hartman.
Thomas M. Riddick's Chapter 14 — " Dynamic Systems "
Static images help us understand, but we live in a Dynamic World & need to view it that way.
Thomas M. Riddick's "Dynamic Systems"– Chap. 14, Part 2 — Asbestos – Blood
This is the part of Thomas Riddick's book that Dr. T.C. McDaniel quotes the most.
Chapter 19 — Dilatancy, thixotropy, and the double and diffuse layers
Chapter 19 explains the science of how agitation can break up blood clots and what else can be done.
Excerpts and Important Material — Glossary
Riddick's Suggested Reference Mateial
The EDTA Story — Using Disodium EDTA as an Anionic Surfactant
by Tommy Cichanowski
Why Animals Don't Get Heart Attacks ... But People Do!
Matthias Rath, MD discusses his studies with Vitamin C and other Nutrients
Animals have an Enzyme in their Livers that makes Vitamin C !!! Lots of it !!!
Vitamin C controls “Free Radicals” a major factor in preventing heart disease.
Dr. T.C. McDaniel — "Using Zeta Potential as a Healing Tool"
The Art of Healing Ourselves
There Has To Be Something Wrong !
A careful look at heavy metal intoxication
by Jann M. Gentry-Glander jmg@derglanderhaus.com
Using Hydroponics to Understand the Earth's Life Processes
On the Atomic Level
Tommy's History Of Western Technology
Site Link List
The Tortoise Shell "Science of Health" Newsletter
— Putting an End to Disease on Our Planet —
Tortoise Shell Life Science Puzzle Box Front Page
View this page Full Frame
 The onset of intravascular coagulation is therefore characterized by a few darting cells in a few capillaries. An increased degree of coagulation is manifested by a greater number of darting cells in a greater number of capillaries. It should be noted that Comets characterize only the first and second degrees of coagulation, becoming less noticeable at higher degrees.
The onset of intravascular coagulation is therefore characterized by a few darting cells in a few capillaries. An increased degree of coagulation is manifested by a greater number of darting cells in a greater number of capillaries. It should be noted that Comets characterize only the first and second degrees of coagulation, becoming less noticeable at higher degrees.






 Diagram showing condition of the blood in the inferior vena cava of the monkey whose blood had been changed to a sludge by Knowlesi malaria. The vessel was between ½ and ¾ inch in diameter. 1. The lowest one–fourth to one–third of the vessel is covered with a paste made up of settled, stationary masses of agglutinated parasitized and unparasitized red cells effectively segregated from the "circulating blood". 2. The middle third consists of somewhat smaller masses carried slowly forward in suspension. 3. The top third consists of supernatant plasma, containing a few small suspended masses all moving forward more rapidly. The flow, if it can be called "flow", of the moving materials was from right to left.
Diagram showing condition of the blood in the inferior vena cava of the monkey whose blood had been changed to a sludge by Knowlesi malaria. The vessel was between ½ and ¾ inch in diameter. 1. The lowest one–fourth to one–third of the vessel is covered with a paste made up of settled, stationary masses of agglutinated parasitized and unparasitized red cells effectively segregated from the "circulating blood". 2. The middle third consists of somewhat smaller masses carried slowly forward in suspension. 3. The top third consists of supernatant plasma, containing a few small suspended masses all moving forward more rapidly. The flow, if it can be called "flow", of the moving materials was from right to left.

 Diagram of rather rigid, elastic masses of agglutinated red cells coming down a stem arteriole and passing out through terminal arterioles. At the upper left, the wall of the stem arteriole resists distention sufficiently so that as each mass passes through it, the mass is forced to distort toward fitting the internal form of the artery. As each mass passes down into the terminal arteriole, it arrives at a point at which the force exerted by the thin–walled terminal arterioze is considerably less than the force exerted by the thicker–walled stem arteriole. Each mass then rounds up by means of its own internal elasticity and approaches a spherical form. Note the three masses farthest out in the portrayed vessels. It is not at all uncommon to observe masses going through these reactions as they pass through the terminal arterioles and pre–capillary arterioles of the bulbar conjunctiva of diseased humans. The significant point here is that the masses have internal elasticity and return toward a spherical form as they pass into the soft–walled vessels.
Diagram of rather rigid, elastic masses of agglutinated red cells coming down a stem arteriole and passing out through terminal arterioles. At the upper left, the wall of the stem arteriole resists distention sufficiently so that as each mass passes through it, the mass is forced to distort toward fitting the internal form of the artery. As each mass passes down into the terminal arteriole, it arrives at a point at which the force exerted by the thin–walled terminal arterioze is considerably less than the force exerted by the thicker–walled stem arteriole. Each mass then rounds up by means of its own internal elasticity and approaches a spherical form. Note the three masses farthest out in the portrayed vessels. It is not at all uncommon to observe masses going through these reactions as they pass through the terminal arterioles and pre–capillary arterioles of the bulbar conjunctiva of diseased humans. The significant point here is that the masses have internal elasticity and return toward a spherical form as they pass into the soft–walled vessels.
 This vessel contained slowly moving settled masses. Note (1) large masses which completely fill the lumen of the vessel, (2) small masses settled between large ones, and (3) that the settled sludge in the upper right portion fell onto the lower internal surface of the vein as it came down around the vertically–placed bend.
This vessel contained slowly moving settled masses. Note (1) large masses which completely fill the lumen of the vessel, (2) small masses settled between large ones, and (3) that the settled sludge in the upper right portion fell onto the lower internal surface of the vein as it came down around the vertically–placed bend.
 Diagram of soft plastic masses passing down an arteriole into terminal arterioles. Note that these masses, unlike those shown in photo to the left, do not round up again as they pass into the narrow soft–walled vessels. These masses are undergoing a plastic deformation.
Diagram of soft plastic masses passing down an arteriole into terminal arterioles. Note that these masses, unlike those shown in photo to the left, do not round up again as they pass into the narrow soft–walled vessels. These masses are undergoing a plastic deformation.
 Cinetracing of a small vein containing. white blood cells in 3 separate types of location; stuck to the wall of the vessel, as described by E. R. and E. L. Clark, entrapped between large masses of settled, stationary erythrocytes located on the bottom of the vessel, and carried freely suspended in the supernatant horizontally moving plasma.
Cinetracing of a small vein containing. white blood cells in 3 separate types of location; stuck to the wall of the vessel, as described by E. R. and E. L. Clark, entrapped between large masses of settled, stationary erythrocytes located on the bottom of the vessel, and carried freely suspended in the supernatant horizontally moving plasma.
 Gravitational separation of settled red–cell masses from suspended circulating white cells. One significant consequence is that any white cell carried along in the flowing supernatant plasma cannot come in contact with, hence cannot ingest, a bacterium deep within the settled stationary blood–cell masses (cinetracing).
Gravitational separation of settled red–cell masses from suspended circulating white cells. One significant consequence is that any white cell carried along in the flowing supernatant plasma cannot come in contact with, hence cannot ingest, a bacterium deep within the settled stationary blood–cell masses (cinetracing).
 A photograph taken by transmitted light through a horizontal microscope, showing some of the settling phenomena in flowing frog blood within a venule approximately 500 µ in diameter.
A photograph taken by transmitted light through a horizontal microscope, showing some of the settling phenomena in flowing frog blood within a venule approximately 500 µ in diameter.
 Gravitational separation of settled red cell masses from suspended circulating white cells. This is a tracing from a motion picture taken through a horizontally aimed microscope. Note the layer of concentrated white cells carried in suspension in the plasma.
Gravitational separation of settled red cell masses from suspended circulating white cells. This is a tracing from a motion picture taken through a horizontally aimed microscope. Note the layer of concentrated white cells carried in suspension in the plasma.
 Same artery as shown in
figure on page 268, seen at a later time. The flow shown here was almost quantitatively separating cell–free plasma from blood cells with just enough plasma between them to lubricate their mutually opposing surfaces and thereby permit forward motion.
Same artery as shown in
figure on page 268, seen at a later time. The flow shown here was almost quantitatively separating cell–free plasma from blood cells with just enough plasma between them to lubricate their mutually opposing surfaces and thereby permit forward motion.










